Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Mergers & Acquisitions
Biobased chemical maker Lygos enters cannabinoid market
by Melody M. Bomgardner
January 16, 2020
| A version of this story appeared in
Volume 98, Issue 3
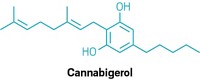
Lygos, a start-up that’s scaling up production of the industrial monomer malonic acid using yeast fermentation, has acquired Librede, a fermentation specialist targeting a very different market: cannabinoids. The two firms have long known each other as members of California’s synthetic biology community, Lygos CEO Eric Steen tells C&EN. He points out that while their markets differ, the purified, rare cannabinoids that Librede makes share structural components with malonic acid. The companies have not disclosed the cannabinoids they plan to sell, but Librede’s process can make common ones such as cannabidiol and tetrahydrocannabinol as well as rare compounds including cannabigerol, cannabichromene, and molecules that have a pentyl group rather than the more typical propyl group. Those molecules, which include cannabidivarin, are hypothesized to have different health effects than their more ordinary cousins. Lygos will apply its expertise in scale-up and purification to make the ingredients for the cosmetics market later this year. That timeline will help Lygos generate sales before it begins building its malonic acid facility in 2022 or 2023, Steen says.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter