Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Medicinal Chemistry
Making heparin safe
Although manufacturing fraud remains possible, Chinese firms have upgraded their processes to meet stricter U.S. regulations
by Jean-François Tremblay
October 10, 2016
| A version of this story appeared in
Volume 94, Issue 40

The pig is king in China. While North Americans and Europeans prefer to eat beef or chicken, pork is the meat of choice in China. In 2014, China slaughtered more than 700 million pigs, roughly one animal for every two people in the country. Pigs are so much a part of the culture in China that the character for home (家) is made up of two symbols meaning pig and roof.
In brief
The 2007–08 heparin scandal caused more than 80 deaths in the U.S. and shook the public’s faith in the safety of drugs, particularly drugs that come from China. Since then, regulators have tightened quality standards for heparin and improved enforcement efforts. Still, questions about heparin’s safety remain. In the pages to follow, C&EN explores the difficulties inherent in manufacturing this unique drug and how one Chinese company is stepping up to the challenge.
China’s huge pig population is the reason why the country is a superpower when it comes to making heparin, an anticoagulant used around the globe during heart surgery and dialysis as well as for the treatment of deep vein thrombosis. The country accounts for half of the world’s heparin production.
Pig intestine mucosa is currently the only approved raw material for producing the heparin sold in most of the world, including the U.S. And given that each mucosa yields only a few grams of heparin, China’s huge pig population is essential to the world’s supply of the drug.
Heparin at a glance
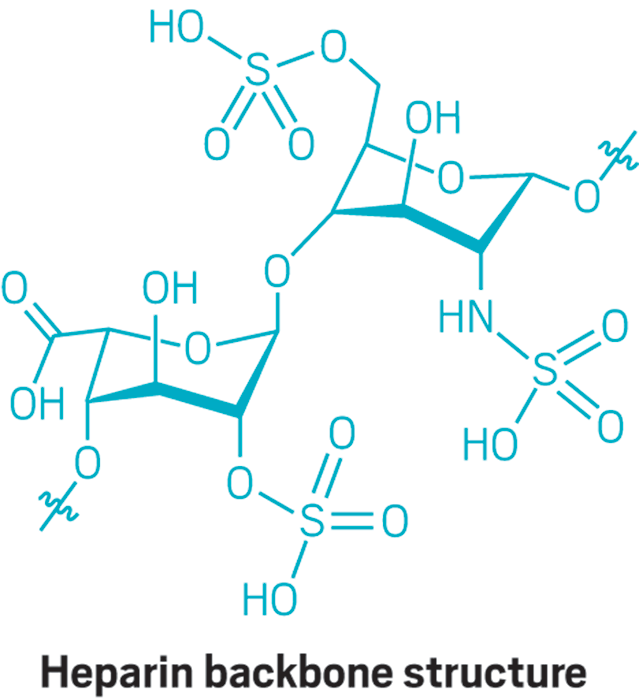
History: Discovered a century ago in 1916, heparin has been used medically since 1935.
Uses: Primarily used as an anticoagulant to treat or prevent deep vein thrombosis or pulmonary embolism, heparin is also used during kidney dialysis. Most heparin is injected into patients in the form of heparin sodium, but companies such as Sanofi have developed drugs made from low-molecular-weight heparin. In the U.S., leading suppliers of heparin sodium include Sagent Pharmaceuticals and Fresenius Kabi.
Production: Heparin has traditionally been extracted from cattle lungs or pig intestines. Since concerns emerged over mad cow disease in the 1980s, cattle in most countries have been banned as a source of heparin. No synthetic heparin is on the market.
Structure: Heparin is a sulfated sugar polymer made up of a sulfated iduronic acid and sulfated glucosamine units.
Ensuring the safety of a substance that is extracted from pig organs thousands of kilometers away poses a great challenge for U.S. regulators, as well as for companies that source heparin to formulate heparin sodium solutions and other treatments based on heparin.
The risks inherent to this long supply chain became apparent in 2007 and 2008 after hundreds of patients in the U.S. and other countries suffered severe allergic reactions to Chinese heparin that had been adulterated with an unapproved additive. More than 80 people died in the U.S. alone.
Nowadays, the production of heparin is subject to far stricter controls than in 2007, making the repeat of such a tragedy less likely. But risks remain, not only because much heparin is from China but also because heparin is a complex mixture of molecules and therefore cannot be characterized exactly. As the U.S. Food & Drug Administration ponders how to further safeguard the safety of the drug, U.S. legislators continue to raise concerns about the quality of heparin made in China.
It may come as a surprise that a drug extracted from pig intestines persists in the era of modern pharmaceutical chemistry. First introduced in 1935, heparin remains an essential drug with unique properties that synthetic anticoagulants such as warfarin cannot replicate.
It may also come as a surprise that, even after the events of 2007 and 2008, the safety of heparin cannot be assured. Back then, the allergic reactions and deaths were caused by heparin from China that had been adulterated with oversulfated chondroitin sulfate, an animal-derived product that at the time couldn’t be detected. Unscrupulous players added the chondroitin to stretch heparin and lower its cost.
Since then, FDA, the pharmaceutical-standards-setting organization U.S. Pharmacopeial Convention (USP), and their counterparts in other countries have sharply tightened heparin manufacturing standards. Among several new measures, FDA inspectors started making surprise visits to Chinese heparin plants. And USP has outlined new limits for common heparin impurities, including oversulfated chondroitin sulfate, and added new tests to verify the animal species the heparin came from.
This tightening of standards has made it far harder to adulterate heparin, but it hasn’t made it impossible. “I believe that poor-quality Chinese heparin nowadays rarely makes its way to the U.S.,” says Zhengjie Mei, founder and chairman of Hubei Enoray Biopharmaceutical, China’s largest producer of crude heparin. “However, some producers still don’t implement the highest standards.”
Among the several Chinese heparin producers that C&EN approached for this article, Enoray was the only one to welcome a visit.

In comments to C&EN, FDA acknowledges limits in its ability to ensure the safety of heparin. “Although FDA cannot prevent or eliminate all possible risks, it can enforce appropriate requirements, controls, and best practices, allowing the early detection of problems and better ensuring the availability of safe and quality medicine,” the agency says. The primary responsibility for ensuring the quality of heparin, FDA adds, lies with manufacturers.

Safety is not easy to ensure for any drug, but for heparin it’s particularly hard. Upstream, the raw material must come from the fresh intestines of healthy pigs. For heparin producers, this means establishing traceability right up to the living animal. And downstream, in the finished drug form, heparin’s bona fides are devilishly difficult to confirm.
In a recent paper, a multidisciplinary group of scientists who developed USP’s updated heparin standards remarked on the difficulty of characterizing heparin (Nat. Biotechnol. 2016, DOI: 10.1038/nbt.3606).

“Although heparin has been used for 80 years,” they wrote, “a simple set of standards for its identity and purity has not existed because heparin is a complex mixture of highly sulfated polysaccharides.” The paper further noted that heparin is “a highly heterogeneous mixture of polysaccharides varying in sulfation patterns and saccharide chain length,” the complexity of the mixture being the result of its origin in animal tissue.
Despite such challenges, Enoray’s Mei is convinced that serious operators can set up manufacturing systems that guarantee the integrity of the heparin supply chain, all the way from the pig to the vial that will be administered to the patient.
Before taking a reporter on a tour of his facilities, Mei explains that he has been in the pig intestine business for 28 years, producing both crude heparin and natural casings for sausages. Enoray started to produce pharmaceutical-grade heparin a few years ago. Earlier this year, the company was acquired by the Tianjin-based finished drug supplier Tianjin Chase Sun Pharmaceutical, which mostly uses Enoray’s heparin to produce low-molecular-weight heparin formulations.

What distinguishes Enoray from other heparin producers is that it controls the supply chain, starting with pig intestines. Many competitors buy crude heparin from third parties, Mei says. The company is located by the Yangtze River near the central Chinese city of Jiujiang. The city may not be one of China’s industrial hubs, but within a two-day drive, Enoray has access to slaughterhouses that can easily supply the 15,000 pig intestines the company processes daily.

Enoray staffers are stationed at the slaughterhouses to approve or reject the intestines before they are loaded onto refrigerated trucks that bring the pig parts to Jiujiang. At the Enoray plant, Mei says, employees also visually inspect the organs and reject any bad ones. “Intestines from sick animals are dark, not bright pink,” he notes.
In the initial stage of processing, operators place the intestines into equipment that separates the mucosa, the inner lining of the intestine, from other intestine components such as the casing material sold to sausage makers. The room where this takes place has the pungent smell of fresh meat.
The mucosa is then piped into large vessels for an enzymatic reaction that separates proteins and impurities from the glycosaminoglycans from which heparin is later extracted. Subsequent operations yield crude heparin powder, which is either sold to other companies or processed into pharmaceutical-grade heparin by Enoray itself.

Each operation conducted by Enoray, all the way back to the slaughterhouse, is documented and individually signed by the operators involved, according to Peter J. Werth, chief executive officer of the U.S. pharmaceutical chemical importer ChemWerth. ChemWerth represents Enoray products in the U.S. and advises the firm on U.S. regulatory requirements. Enoray passed a surprise inspection by FDA inspectors last year, Mei says. Werth expects that the firm’s heparin will be available to finished drug formulators in the U.S. in a matter of months.
During the plant tour, Mei points to an operator who is conducting polymerase chain reaction testing on crude heparin before it is purified into the pharmaceutical grade.
The PCR test is a new FDA requirement that verifies the animal species from which the heparin is extracted. Although some countries allow other animal sources, only porcine heparin is permitted in the U.S. FDA started requiring the test in 2013 to reduce the risk that heparin from sheep, goats, or cattle enters commerce as an active pharmaceutical ingredient, or API. Bovine heparin was discontinued in the U.S. in the 1990s owing to concerns over mad cow disease, and sheep and goat heparin were never tested on humans. “Once the heparin is in API form, it’s impossible to determine which animal it came from,” Mei observes.
Some heparin from sheep or other unapproved animal sources could still be entering the supply chain. On commercial websites in China, it is relatively easy to find advertisements for crude sheep heparin costing far less than the porcine product.
In 2015, French regulators discovered that China’s Dongying Tiandong Pharmaceutical had manipulated crude heparin PCR data indicating the presence of “ruminant” sources, meaning cattle, goats, or other grass-eating animals. Fudged data in hand, Tiandong processed the crude heparin into purified API and sold it as porcine-derived.
Manipulating PCR data may not be the only nefarious activity engaged in by dodgy heparin makers. In early 2014, the heparin producer Beijing Shunxin Meihua Bio-technical simply barred FDA officials from entering its facilities. FDA issued a warning letter to the firm and banned its products from the U.S.
Members of the House of Representatives Committee on Energy & Commerce continue to worry about the possible presence in the U.S. of heparin from nonporcine or adulterated sources. In March, they sent a letter to FDA Commissioner Robert M. Califf highlighting their concerns about the heparin supply chain.
Perhaps their most damning claim was that China just does not have enough pigs to make all the heparin it produces.
Chinese regulators require that heparin be extracted only from intestines produced at regulated slaughterhouses. In 2008, such facilities accounted for just more than half of the pigs that China butchered. Using Chinese heparin export statistics and the rule of thumb that it takes 2,000 pig mucosae to produce 1 kg of heparin, the legislators concluded that “there is an inherent shortage in the legal pig supply in China to make heparin.”
U.S. concerns over the quality of Chinese heparin are starting to be unreasonable, argues Ruixin Miao, head of Nanjing Kaiyang Biotech, a Nanjing-based heparin trading firm. “The new FDA standards make it very hard to adulterate the product or use other animal sources,” says Miao, who has been involved in the production and trading of heparin for decades.
China, Miao adds, has more than enough pigs to supply its own heparin needs as well as the export market. Owing to productivity improvements, it now takes about 1,500 intestines to produce 1 kg of heparin, not 2,000 intestines as U.S. lawmakers have claimed. And unlike in 2008, most major slaughterhouses are now overseen by the government, he says, meaning that far more than half of slaughtered pigs are available to heparin producers. “The small slaughterhouses, by definition, they don’t slaughter a lot of pigs,” he notes.
Even if only 300 million pigs are available for heparin in China every year, that’s enough to produce 17.6 million megas of heparin, Miao says, referring to a measurement unit used for heparin. “World demand is 28 million megas, and the rest of the world outside China produces 14 million units. So China has spare production capacity.” Europe is the world’s second-largest heparin producer, accounting for 8 million megas, Miao estimates.
With the beefed-up FDA requirements, it is almost impossible to contaminate heparin with oversulfated chondroitin sulfate, Miao argues. Using other animals remains a vulnerability, he concedes, but all in all, raising compliance requirements much further could raise costs to the point that heparin shortages would start to occur, he says.
Shortages nearly occurred in 2008 when numerous batches of contaminated heparin were recalled in the U.S. and Europe. Looking ahead, FDA has been mulling reintroduction of heparin extracted from cattle “to address such issues as possible shortages and economically motivated adulteration of porcine heparin,” the agency tells C&EN. Bovine heparin was available in the U.S. until the 1990s, when it was discontinued over concerns about bovine spongiform encephalopathy (BSE), or mad cow disease. Bovine heparin is still sold in Brazil, a major cattle producer.
Advertisement
FDA says it is working with USP and foreign regulatory agencies, as well as heparin experts, to develop new standards to characterize and quality test heparin from bovine sources. The agency notes that the risk posed by BSE can be minimized by using cattle raised in countries free of the disease and by using only the animals’ lungs and intestines.
But some scientists contend that reintroducing bovine heparin could compromise heparin safety. “You can expect more unwanted side effects in patients if you use heparin from bovine sources,” says Guangli Yu, a professor in the School of Medicine & Pharmacy at Ocean University of China who studies heparin. Pigs are more genetically similar to humans than cattle are, Yu says, so porcine heparin poses less of a risk of allergic reaction.
“If you want to improve the quality of heparin, turning to another species may not be the right solution,” he says.
Bin Liu, the sales director at Enoray, points out that “bovine heparin may help cut out China as a source of supply, but if you get the threat of BSE in return, you’re not that much ahead.” In addition, he notes that all scientific research on heparin since the 1990s has been based on heparin from porcine sources. It will take a lot of work, and new clinical trials, to determine when and how bovine heparin can perform like the porcine version.
To get around the problem that sourcing heparin from animals poses, one solution could be synthetic heparin or a synthetic drug that performs like heparin. Several groups of researchers around the world are trying to do just that, despite the long odds against succeeding.
“When we first started, we tried to make heparin itself,” recalls Jian Liu, a professor at the Eshelman School of Pharmacy at the University of North Carolina, Chapel Hill, who heads a group that has been trying to synthesize heparin for more than a decade. “But how can you reproduce a molecule that cannot be precisely characterized and that also contains 40 sugars?”
In recent years, Liu’s group has had some success trying to reproduce fragments of heparin containing about a dozen sugars. The group focuses on fragments that have medical activity and that would, in the end, resemble drugs such as Sanofi’s Lovenox that are based on low-molecular-weight heparin. Liu is confident that his group will have a clinical drug candidate within a couple of years.
“We are able to control the size of the molecule so that it can be metabolized in the kidney or the liver, depending on the patient’s condition,” Liu says. His group has also come up with an agent that can reverse the effect of the drug for patients who respond negatively.
While researchers such as Liu continue to explore synthetic versions of heparin, Werth, the API importer, is gearing up to introduce Enoray’s pig-derived heparin to the U.S. Because his firm advises Enoray on regulatory compliance and audits its quality-control systems, Werth is confident that quality will not be an issue.
But he cannot vouch for other companies, even with the higher standards that FDA has imposed in recent years. The problem, he says, is human nature. If there is a way to make more money by adulterating a product, some will be tempted. “You can only take it so far,” he says. “Somebody who wants to cheat will find a way.”



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter