Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Pharmaceuticals
Continuus Pharmaceuticals
Improving efficiency with end-to-end drug manufacturing
by Ann M. Thayer
October 31, 2016
| A version of this story appeared in
Volume 94, Issue 43
Continuus Pharmaceuticals
Launched: 2013
Headquarters: Woburn, Mass.
Focus: pharmaceutical manufacturing
Technology: continuous production from synthesis through finished dosage form
Founders: Salvatore Mascia and Bayan Takizawa
Funding or notable partners: spin-off of a $65 million collaboration between MIT and Novartis
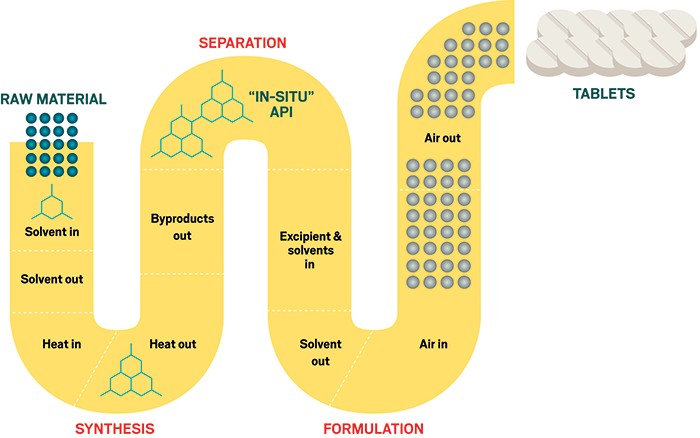
Continuus Pharmaceuticals has hit all the traditional milestones for a young start-up: university spin-off, seed funding, business incubator, small-business grants, Series A investment, and a move to a new facility. Now it must convince the pharmaceutical industry to make a technological step change in how it manufactures its products.
For decades, drug companies have used discrete steps and equipment, often housed in multiple locations, to make their active pharmaceutical ingredients (APIs) and finished products. One industry analysis estimates that the pharma industry wastes $50 billion a year on inefficient processes. To stem this loss, Continuus is offering to design new facilities that can go seamlessly from raw materials to drug tablets.
“Our vision is to help the industry transition from outdated batch manufacturing methodology to continuous manufacturing,” says Bayan Takizawa, Continuus’s cofounder and chief business officer. Doing so, he says, can reduce lead times, equipment size, environmental impact, and costs. In addition, production can be more flexible and more reliable through better process and quality control.

The Continuus team, including founder and CEO Salvatore Mascia, gained experience within the Novartis-MIT Center for Continuous Manufacturing. In 2011, when Mascia was its strategic project manager, the $65 million center unveiled a proof-of-concept pilot plant. Its nonstop process incorporated synthesis, separations, purification, crystallization, drying, formulation, and tableting in one stream.
Since then, Novartis has implemented continuous processes at its facility in Switzerland. Continuus was spun off in 2013 to bring the technology to the broader market. Its clients, Takizawa says, now include innovator drug companies as well as generics firms.
The start-up also signed up strategic partners, including the Italian pharmaceutical equipment supplier Industria Macchine Automatiche (IMA), which is Continuus’s lead investor as well. New England Controls, an affiliate of Emerson Process Management, provides automated control systems.
With 10 full-time staff members, Continuus has begun developing routes to specific compounds. The plan is to eventually scale up such systems and either sell or lease them for use at a customer’s facility or operate them under contract, Takizawa says.
Continuus isn’t the first to offer continuous processes. Contract API manufacturers such as Lonza and SK Biotek are developing flow chemistry reactors, and drug formulators are starting to employ continuous crystallization and tableting. But few firms are integrating manufacturing from end to end.
As a result, the biggest challenge isn’t competition, Takizawa says, but getting potential customers to think about new ways to make products and then consider Continuus’s approach.
Some big firms are already moving in this direction on their own. Besides Novartis, Eli Lilly & Co. and GlaxoSmithKline are installing continuous production lines. And dosage forms of Vertex Pharmaceuticals’ cystic fibrosis drug Orkambi and Johnson & Johnson’s HIV drug Prezista are made via continuous methods. Both drugs are signs that regulators support the new approach.
“Over the next year or two, we hope to continue to show the benefits of our technology and begin work with other pharmaceutical clients,” Takizawa says. “We are also staying ahead of the curve and developing new technologies that will keep the field of continuous manufacturing evolving.”

Know a start-up we should consider for next year's list? Nominate it here: cenm.ag/startupnom





Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter