Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Infectious disease
Covid-19
Podcast: Why do I have to get a flu shot every year?
Influenza evolution experts tell Stereo Chemistry what makes the virus so good at evading our immune system
by Bethany Halford
January 31, 2020
| A version of this story appeared in
Volume 98, Issue 5

Although the Wuhan coronavirus is dominating headlines across the globe, influenza kills hundreds of thousands of people worldwide each year. In the US, millions of people roll up their sleeves annually for a flu shot. But this ritual is confusing for many. Why is it that most vaccines are effective for a lifetime while the flu vaccine is only effective for a year? And why do we sometimes get the flu even when we’ve gotten the vaccine? The answer is evolution: the flu is constantly evolving to evade our immune systems. In this episode of Stereo Chemistry, scientists who study flu evolution and pandemics explain what makes fighting the flu so difficult.
Subscribe to Stereo Chemistry now on Apple Podcasts, Google Podcasts, or Spotify.
The following is the script for the podcast. We have edited the interviews within for length and clarity.
Support nonprofit science journalism
C&EN has made this story and all of its coverage of the coronavirus epidemic freely available during the outbreak to keep the public informed. To support us:
Donate Join Subscribe
StefanieOlsen: This is the info sheet from the CDC on the flu vaccine. Kind of who should get it, why you should get it, who shouldn’t get it, what to expect, what’s normal, what’s not normal. All that sort of stuff. So I’ll give you that for your perusal.
Matt Davenport: That’s Stephanie Olsen. She’s a nurse practitioner at a MinuteClinic in Cambridge, Massachusetts. That’s where C&EN senior correspondent Bethany Halford and her son went to get the flu vaccine back in the fall.
Stefanie Olsen: Are you a righty or a lefty?
Bethany’s son: I’m a righty.
Stefanie Olsen: OK. Cool. We’ll use your left arm. Find this big muscle. Here we go: clean, clean, clean. OK. One, two, three. Good job. Done. There you are.
Bethany’s son: One tiny sting.
Stephanie Olsen: One tiny sting and done. Good job.
Matt: That didn’t seem so bad.
Bethany Halford: It really wasn’t bad at all.
Matt: Well hello there, Bethany.
Bethany: Hey, Matt.
Matt: Thanks so much for bringing your recorder along with you for the flu shot.
Bethany: No problem. I’m actually glad I made this recording because I plan to replay it for my son every year just before we go to get our shots. It’s a process that’s met with no small amount of dread. But the Centers for Disease Control and Prevention recommend that most people get the flu vaccine every year.
Matt: So you and your son went in September. It’s now almost February. Let’s pretend you’re a podcast cohost who has not gotten their flu shot. Is it too late?
Bethany: Well, CDC does recommend getting the flu vaccine by the end of October because it takes a few weeks for your body to create the antibodies that fight the virus. And this year the flu seems to be ramping up early. But doctors say that even now, it’s not too late to get the vaccine.
And we’re right in the thick of flu season. During the last flu season in the Northern Hemisphere, from October 2018 to May 2019, as many as 42.9 million people in the US got sick with the flu; 647,000 of those people were hospitalized, and 61,200 died.
Matt: Those numbers are from CDC, and they’re pretty typical for a flu season. So influenza is this huge problem, and it’s been that way for a long time. And it’s not going away, right? Unlike other vaccines, the flu shot is something you should get every year. And sometimes that flu shot isn’t going to work.
Bethany: And this episode is all about how the flu outfoxes our vaccines and immune systems: through evolution. The flu virus is constantly changing itself to evade our immune system’s response. And the virus changes enough each year—sometimes even enough within a single flu season—that the vaccine we’ve created is simply no longer effective.
Matt: So Beth, at the risk of sounding like a chemist right after the Nobel Prize announcement, isn’t that a little more biology than chemistry?
Beth: Well, yes. But the evolutionary changes to influenza are really chemical changes. They’re mutations in the virus’s RNA that lead to amino acid changes in the virus’s proteins. So there is plenty of chemistry to dig into. We’re going to talk to three experts to learn how those changes happen and how studying them could help protect us better in the future. We’ll also look at what happens at the molecular level when a certain strain of flu becomes a pandemic that spreads quickly across the globe.
And we’re going to start by talking to a chemist.
Jesse Bloom: Hi, my name is Jesse Bloom.
Bethany: Jesse studies protein evolution at the Fred Hutch Cancer Research Center in Seattle. He’s also affiliated with the University of Washington and the Howard Hughes Medical Institute.
Jesse Bloom: I actually did my PhD in chemistry, working with Frances Arnold, who studied the directed evolution of proteins.
Matt: Wait. The Frances Arnold?
Bethany: Yes, the Frances Arnold from Caltech who won a share of the 2018 Nobel Prize in Chemistry.
Jesse Bloom: After working with Frances, I remained really interested in protein evolution, but I wanted to study the evolution of proteins in a context with biomedical significance. So my lab now focuses on viral evolution, particularly the evolution of influenza virus. And the reason for that is these viruses evolve their proteins very rapidly.
Bethany: Jesse says there are really two main forms of flu evolution. One is called antigenic drift, and the other is called antigenic shift.
Matt: I like the rhyme scheme.
Bethany: Catchy, right? So let’s start with the drift.
Jesse Bloom: Antigenic drift is the much more common form of flu evolution, and that essentially can be thought of as last year’s strain or a couple years ago strain of human flu evolving to be a little bit different, each year. Our immune systems are actually great at mounting antibody responses that protect us against flu, and there’s pretty good evidence that if you’re infected with a particular strain of flu, your body will provide very good, long-lasting immunity to that particular strain of flu.
Bethany: So, if our bodies provide long-lasting immunity, I’m sure you’re wondering why we still have to get a flu shot every year.
Matt: You are correct.
Bethany: Here’s how Jesse explains it.
Jesse Bloom: The challenge with flu is the virus evolves very rapidly. In particular, the positions on the viral proteins that are recognized by our immune system, primarily by our antibodies, change, and they change enough that after about 5 years, many of those antibodies sort of don’t work anymore. So antigenic drift and what typically is responsible for the seasonal influenza outbreaks is the virus that was present last year or the year before changing a little bit so that after about 5 years, it’s mostly evaded your immune system’s memory.
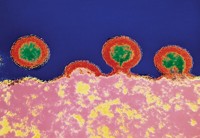
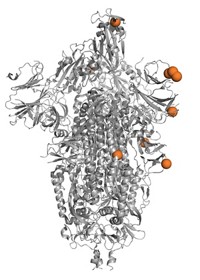
Bethany: Now, I told you this was a chemistry story, so before we go any further, let me give you a picture of what Jesse is talking about. There are two proteins that scientists think are most important with respect to immunity—we create antibodies that bind to these two proteins in order to mount a defense against influenza. The first protein is hemagglutinin, which helps the influenza virus latch on to cells and infect them. The second is neuraminidase, which helps cleave new virus particles away from infected cells so the virus can continue to attack healthy cells. If you think of the flu virus as a sort of blob, hemagglutinin and neuraminidase stick out of that blob like pins in a pin cushion. Scientists name different strains of flu based on which types of hemagglutinin and neuraminidase they have.
Matt: Are those the proteins we’re referring to when we talk about like H1N1 influenza or H3N2 influenza ?
Bethany: That’s right. Right now, there are three types of flu circulating in humans: H1N1, H3N2, and influenza B.
Jesse Bloom: I mean, they all evolve pretty fast, like, compared to almost anything else we encounter in life. But definitely H3N2 evolves the fastest. H1N1 is sort of in the middle. And influenza B is the slowest, although influenza B is still pretty fast. And this plays out—for instance, influenza B is most known for infecting children because it’s relatively less good at escaping immunity. Obviously children don’t have any immunity at all, if they haven’t been vaccinated, anyway, to escape. So they’re always going to be susceptible. And then H3N2 is sort of best at infecting older people—it’s also good at infecting younger people, but it’s good at affecting all ages—and probably the reason is that H3N2 is evolving the fastest. So it can best get away from that prior immunity.
Matt: So, when he says something is evolving fast, what does that mean on a molecular level?
Bethany: Take H3N2 influenza, for example. The hemagglutinin protein on H3N2 will change three to four of its amino acids every year—an evolution rate that Jesse says is extraordinarily high.
Matt: OK, so I understand why these gradual changes—the antigenic drift—make it so that we have to get the flu vaccine every year. But why don’t we need frequent vaccinations for all RNA viruses? Like measles?
Bethany: CDC recommends just two shots for measles as part of what’s called the MMR vaccine. It protects you from measles, mumps, and rubella. You get the first shot when you’re about a year old, the other when you’re about 5 years old. It seems that the parts of the measles virus that the immune system goes after—or makes antibodies for—just don’t seem to be changing that much. We know this because before the measles vaccine existed, people who got measles only got it once in their lifetime. And in the 50 or so years since we’ve had the vaccine, people who get it don’t get measles. As Jesse explains, there’s no reason measles can’t drift like the flu, that’s just not what we see. So the thinking is that measles is mutating, but not in a way that helps the virus. It’s not as wily as influenza.
Matt: That is super interesting. But . . .
Bethany: How does knowing this help fight the flu?
Matt: Yeah.
Bethany: I asked Jesse.
Bethany (in interview): Can you talk a little bit about how studying flu’s evolution can help us fight the virus?
Jesse Bloom: So first, the way the flu vaccines are made currently, there’s sort of this forecasting problem. We know that the vaccine works better when the vaccine is more similar to the virus that is infecting people. But it takes a while, maybe about 9 months, to really produce enough vaccine to be given to everybody. And because the virus is changing a little bit every year, you have to predict what virus is going to be circulating 9 months in the future. So you basically have to say, “How do we think the virus is going to be evolving?” And so by understanding the virus’s evolution, we can make better decisions about which flu strain should go in the flu vaccine. And when those decisions are better, the vaccine will work better.
Bethany (in studio): Jesse also says that studying evolution helps scientists understand which parts of the flu virus change the least or mutate less frequently. It could be that some of these less-dynamic parts of the flu could become targets for longer-lasting vaccines.
Matt: I can dig it. So what’s driving the evolution? What’s making the proteins change?
Bethany: Good question. Let’s get another influenza evolution expert to chime in.
Adam Lauring: So I’m Adam Lauring. I’m an associate professor here at the University of Michigan. I am a physician-scientist, which means I spend part of my time actually doing clinical work in infectious diseases. But most of my time I spend actually running a research lab, in which we study virus evolution, including influenza virus. Evolution is really for me kind of the be all, end all in the problem of influenza. Evolution has immediate and real-world impacts.
Advertisement
Bethany (in interview): When we say flu is evolving, what is actually going on?
Adam Lauring: At its simplest, the flu will mutate, and that means that it’s making changes in its genome which will lead to changes in its proteins, and those protein changes will make the virus different. And then there’s selection. And so viruses that are better at doing what viruses do will take over, and the viruses that are less fit will die away. And so it’s kind of like you learned when you first learned biology: it’s survival of the fittest, or the best one wins. And so the virus is mutating all the time, and the ones who are best able to make copies of themselves and spread from person to person are going to become the new viruses and replace the old ones.
Bethany (in studio): Now, flu evolution is a complex process that’s influenced by many things. But one thing that helps flu evolve especially fast is that it’s an RNA virus. That means its genes are stored in ribonucleic acid, or RNA. RNA viruses, in general, evolve faster than viruses that store their genetic information in DNA. Both DNA and RNA viruses have proteins called polymerases, and the job of these proteins is to make copies of the virus’s genetic code. DNA polymerases, however, have a built-in proofreading function. They can check their work for mistakes and correct them. RNA polymerases don’t do that.
Adam Lauring: Because of this, most RNA viruses have mutation rates or error rates that are about a thousandfold higher than for DNA viruses. That means that an RNA virus can generate mutants way more quickly, and then some of those mutants will confer an advantage to the virus, and that will lead to faster evolution.
Bethany: Adam says that all of the flu virus’s proteins can and are evolving but that mutations to the hemagglutinin and neuraminidase proteins—the Hs and Ns—are the ones that matter most.
Adam Lauring: Mutations in those proteins tend to make a bigger difference in terms of whether the virus succeeds or fails, and a major reason is those proteins, they’re on the surface of the virus, and so they’re targeted by the immune system. And so you have antibodies targeting those proteins. So if a virus figures out a way to escape those antibodies, it will do better than its brothers and sisters.
Bethany: So we’ve been talking a lot about mutation, but Adam also points out that there’s a lot more to evolving quickly than just how fast a virus mutates. For instance, the number of people infected could play a role. The example he gave me is the more people infected, the more opportunities the virus has to evolve. That’s because a greater diversity of people would mean a wider variety of immune systems, and the virus would need to generate new or different versions of itself to survive.
Adam Lauring: Broad strokes, flu does evolve quickly but maybe not for the reasons we typically think. And there are probably subtleties yet to be uncovered.
Bethany: To try to uncover some of those subtleties, Adam’s lab has been collaborating with Arnold Monto and Emily Martin, who are epidemiologists at the University of Michigan School of Public Health. For about 8 years, they have been following 300 or so Michigan families to see what viruses are circulating among them and how their immunity changes over time. The flu virus is part of this sampling. As part of the work, they collect nose and throat swabs anytime someone from one of those families gets sick.
Matt: Oh, wait. Everyone gets swabbed when anyone gets sick?
Bethany: Right. Here’s why.
Adam Lauring: It’s really kind of a slice of what flu is doing locally, and you’re not really biased by only getting sick people or people who tend to go to the doctor.
Bethany: Adam’s group realized that the collection of samples the epidemiologists had accumulated gave them a great opportunity to see how flu viruses were evolving outside of a laboratory. So they raided the freezer and then did in-depth genetic sequencing of all the influenza viruses they found.
Adam Lauring: The virus makes a lot of mutations. Everybody’s flu viruses, their population is actually a little bit different. So I could have the flu and you could have the flu and we’d be in the same room, but our flu viruses might be a little bit different if you really looked hard enough. And so what we’re able to do with our sequencing is really understand those subtle differences in kind of the overall flu mixture that each person has in them.
Bethany: And then they compare, see which versions are actually being transmitted from person to person.
Adam Lauring: And that is really important in understanding evolution, right, because you may generate all sorts of cool viruses inside you. But if they don’t make it onto the next person, it’s kind of a dead end. And that virus could be the most awesome virus there is, but if it doesn’t get transmitted, it’s gone forever. And so what we tried to do is understand exactly how many viruses kind of go across from one person to the next. And we found that it’s actually a really small number. It’s hard for a new virus to kind of make it both within a host and to get on to the next host.
Matt: That’s wild. So, if it’s hard for a new flu virus to survive within a host and also hard for that virus to make it to the next host, how is that much evolution happening? Why do we still need to get the flu vaccine every year?
Bethany: Adam says it’s really just a numbers game. Hundreds of millions of people are infected with the flu each year, which gives the virus lots of opportunities to make a successful mutant.
Adam Lauring: One analogy I give is flu viruses are sort of like people playing the slot machines. And so most of the time the virus is losing when you talk about kind of on an individual host or in a household. But if you have a hundred million people playing the slot machines, you’re going to hit the jackpot with some frequency.
Matt: I like that analogy. It’s kind of empowering. Like humanity’s the house and the flu’s a rube giving us their money.
Bethany: Sure. Just remember, the flu’s currency isn’t money. It’s trying to survive, and when it thrives, it makes you sick. So it’s not like a casino catches fire whenever someone hits the jackpot. And the analogy really works best for antigenic drift. We’ve got a whole other type of evolution to talk about—remember how I said there were two? This second kind leads to pandemics, and we’ll talk about it . . . after the break.
Matt: Hey. Sorry to leave you hanging like that, but don’t worry. There’s going to be a silver lining. We’re not just going to be like. “The flu. Yeah, it’s brutal. Welp, see you later.”
That’s the great thing about covering chemistry. It’s that we’re not just talking about problems, we’re talking to the people solving them.
In fact, earlier this month, Leigh Krietsch Boerner wrote a phenomenal piece for C&EN about how researchers are examining the effectiveness of flu shots, especially vaccines made using eggs.
We’ve got a link to Leigh’s story in the description, but if you want to inoculate yourself against the possibility of missing more of our great coverage, sign up for our newsletter. We’ll send a weekly dose of chemistry’s biggest goings-on right to your inbox. Head to cenm.ag/newsletter to subscribe.
Matt: So, Bethany, you said there were two main forms of influenza evolution: antigenic drift, which we’ve been talking about. But there was also, what was that rhyme again?
Bethany: Antigenic shift.
Matt: Right, antigenic shift. What’s that?
Bethany: When the influenza virus undergoes antigenic shift, it experiences a much larger change. It changes so much, in fact, that we usually don’t have much of an antibody arsenal built up to fight it.
Matt: And how does it make such a dramatic shift?
Bethany: So, antigenic shift can happen a few different ways. Another way flu is different from measles is that flu doesn’t just circulate in people. It also circulates in many other animal species, like pigs and whales and birds.
Matt: Whales?
Bethany: Yeah. But it turns out, the vast majority of influenza strains that exist in the world actually are circulating in wild waterfowl. And sometimes those viruses will jump from birds to people or from birds to pigs to people, for example.
Advertisement
A single animal can also get infected by two different strains of flu from two other animals. Those viruses then swap some of their genetic material to make a new, third strain.
However it’s happening, when the flu is evolving outside of humans, vaccine makers and our immune systems are largely blind to what these viruses look like. That means if one of these viruses does jump to humans, it could hit us hard. We’re talking global pandemic here. That’s because the virus would look very different from anything our immune systems have seen, and we might have little or no ability to recognize the strain or fight it.
Matt: That sounds gnarly. And a little scary.
Bethany: It is. Global flu pandemics occur when a novel influenza virus spreads quickly around the globe.
Matt: Is that why we’re so concerned when people get infected with flu on chicken farms, for example?
Bethany: Yes. And you may have heard about the recent outbreak that started in Wuhan, China. That’s a coronavirus—so, not the flu—but it’s another example of a pathogen that made the jump into people from animals. But there’s actually a lot more to becoming a global pandemic than just generating a virus people haven’t seen before. Let’s talk to someone who studies how global influenza pandemics emerge.
Seema Lakdawala: My name is Seema Lakdawala. I am an assistant professor at the University of Pittsburgh in the School of Medicine and the Department of Microbiology and Molecular Genetics.
Bethany: Seema says there are several hurdles a new virus has to overcome before it can become a pandemic
Seema Lakdawala: And the first hurdle is that they have to be able to infect the human host. And so it’s hard for some viruses that may be emerging in birds to infect the human hosts unless there’s access. And so it doesn’t happen as readily, but that does happen in many occasions.
Matt: That makes sense, right? It’s kind of like what Adam was talking about earlier. How you can have all these cool bugs being made in humans, but if they can’t survive, and if they can’t make the leap in humans, they really aren’t a threat.
Bethany: Right. And Seema says the next hurdle, after a virus has made it into a human, is the virus being able to survive in respiratory systems. In humans, the flu is a respiratory infection, but in birds, it’s gastrointestinal. Influenza virus can move from birds to people through contact with feces or other secretions, but—don’t worry—not from eating poultry or eggs.
Still, the virus has to evolve so that it can live in the human respiratory tract. Then it has to be airborne transmissible. It needs to be able to move through the air from person to person to infect human airways.
Seema Lakdawala: And this is actually a really high bottleneck and that a lot of viruses that are able to jump species, from like birds into humans or pigs into humans, are not capable of really sustained human-to-human transmission.
Bethany: But when a novel flu virus makes it over all these hurdles, we see a global flu pandemic. And these seem to happen every few decades.
Matt: So I remember the 2009 swine flu pandemic. But there were others in relatively recent history?

Bethany: That’s right. There was also one in 1977, one in 1968, and one in 1957. Then, of course, the famous 1918 flu pandemic.
Matt: The Spanish flu.
Bethany: Yeah. More people died from that pandemic than died as a result of World War I. There were about 20 million military and civilian deaths from the First World War. But between 50 and 100 million people died during the Spanish flu epidemic, which scholars believe actually started in Kansas. And this brings us to another reason researchers are studying flu evolution. If we can understand the molecular history of pandemic viruses, we have a better chance of preparing ourselves for the future.
So let’s look a little bit at the biochemistry of the 1918 flu. It was an H1N1 strain. And the 2009 swine flu strain was also an H1N1 strain. Now, usually different strains of H1N1 flu are pretty different—like, as different as a human and mouse. But Seema tells us that these two H1N1 strains from 1918 and 2009 are actually pretty closely related.
Matt: Whoa. How is that possible?
Bethany: Well, scientists think that back in 1918, the same virus that jumped into humans also jumped into pigs. And while the human virus kept drifting until it became something very different, the virus in pigs didn’t evolve all that much.
Matt: So the version hanging out in pigs shifted back into humans to become the swine flu of 2009.
Bethany: Kind of. Do you remember Jesse Bloom, the researcher who studies protein evolution we heard from earlier in the episode?
Matt: I do.
Bethany: He’s got the molecular details on how relatives of the 1918 pandemic helped create another one in 2009. But for those to make sense, you also need to understand a couple of different timelines—namely, the average lifetime of a human and the average lifetime of a pig.
Jesse Bloom: Most humans who are infected with flu have already been infected in the past because humans live for a long time. And so that basically means in order for the virus to keep reinfecting humans, it has to evolve to get away from this immunity. Most pigs, particularly pigs on commercial swine farms, don’t live very long. And so often pigs that are infected are naive. So the virus doesn’t have to sort of evolve to escape from the immunity. So the hemagglutinin in pigs change much more slowly. So in 2009, hemagglutinin was actually sort of the pig descendant of the hemagglutinin from 1918. So it certainly wasn’t the same hemagglutinin, but it was similar in that it had just been evolving in pigs so it had been changing more slowly.
Matt: So let me see if I get this. The 2009 swine flu contained hemagglutinin that had been circulating in pigs since 1918 without changing much and then it jumped into people. That is wild.
Bethany: It is. And it gets even wilder. There was this really peculiar pseudopandemic in 1977 that Jesse Bloom told me about.
Jesse Bloom: So that was a pandemic that primarily affected people who are younger than about 25 years old or 24 years old. So one of the chemical concepts which has been very important in evolutionary biology is the idea of a molecular clock. So this was actually an idea that was originally proposed by Emil Zuckerkandl and Linus Pauling. They were at Caltech, and what they noticed is that when you look at the evolution of any protein, you can see the protein changes at sort of a constant rate. And so they called this a molecular clock. And so normally when you look at flu viruses, you can sort of see each of the proteins changing at sort of a constant rate, a couple of mutations every year. But this virus, which appeared in 1977, looked exactly like the virus that had been present in 1953 or 1954, so its molecular clock had frozen.
Bethany: And when he says frozen, he means literally.
Jesse Bloom: So basically, that virus must have been existing somewhere where it wasn’t undergoing evolution. And since flu viruses don’t have the ability to, like, go dormant. They’re not very physically stable. That almost certainly means it was frozen somewhere, and the belief was actually it was probably a virus that was frozen in some sort of laboratory freezer and then was inadvertently rereleased into the human population.
Bethany: The reason only younger people were affected is because those who were older had already been exposed to the virus.
Matt: That is straight up bonkers. I mean, like, terrifying.
Bethany: It is. But remember, researchers want to know where these particularly nasty strains come from so we can protect ourselves better in the future.
Jesse Bloom: We want to understand where they came from. So we can understand where sort of new future outbreaks may come from. And one of the things that evolutionary biology is very good at now is looking at the sequences of viruses that exist and essentially sort of building family trees that allow us to say, Where did these viruses originate? So by studying virus evolution, we can understand a lot about how the viruses changed in the past, which helps us have insight into what might cause new epidemics and pandemics.
Matt: That is reassuring, but still, does all this talk of influenza pandemics make you nervous?
Bethany: To be honest with you, Matt, it really does. That 1918 flu pandemic was just so devastating, and it was only about 100 years ago. But I asked each of our flu experts if they worried about flu pandemics, and they have a different view. Here’s how Michigan’s Adam Lauring looks at it.
Advertisement
Bethany (in interview): As a flu researcher, is the flu or flu pandemic something you worry about? Is it something that keeps you up at night?
Adam Lauring: No, I sleep well. Of course the big concern is, like, would we have something like 1918 again. That would keep me up at night. And I guess I just thought that that’s . . . I think things are very different in important ways from 1918 in that 1918 we didn’t have modern health care. There were no vaccines. Even though, you know, a vaccine might not work against a pandemic strain, it might provide enough protection such that it wouldn’t cause as big a problem. We have antibiotics that we didn’t have in 1918 and most people in 1918 died of pneumonia, bacterial pneumonia which came after the flu. And so maybe the 2009 pandemic would have been a lot worse if we didn’t have antibiotics and modern health care. And so that makes me think that will tend to make things not as bad as they were in 1918.
Bethany (in studio): And Seema Lakdawala at Pitt agrees.
Seema Lakdawala: That is one aspect that we cannot ignore, right, that we are just as a society better prepared for public health burdens. It doesn’t keep me up at night. I do like thinking about what I think the next pandemic is going to be and making sure, if that’s what it is, how could we be prepared? And thinking about ways that we could have more universal precautions. To me, that’s why I really love to study transmission is because if we can stop a virus from transmitting, the threat of a pandemic becomes almost negligible, right. Like we can just stop it. We don’t have to worry about having vaccines stockpiled against certain subtypes of flu that we’re finding in a bird somewhere. If we can just block it, then fantastic. We will be able to really minimize the severity of a pandemic. So, yeah, no, it doesn’t keep me up at night. But it’s something I enjoy thinking about. So hopefully that doesn’t sound too weird to the audience.
Matt: OK, so what about Jesse Bloom?
Bethany: He’s also not worried, but in a worrying kind of way.
Jesse Bloom: I wouldn’t say keeps me up at night. I mean, I have a, like, 21-month-old. I’m usually pretty tired. Not much keeps me up at night at this point. But it’s something that I definitely worry about. I mean, we know that flu pandemics move around the world very quickly, even in 1918, when many forms of transportation in the world moved a lot slower than they do now. I mean, you couldn’t get from Beijing to New York in 12 h or whatever back in 1918. But still, that virus spread around the world quite rapidly and killed a very large number of people. And there’s no reason that something like that couldn’t happen again. So I do think that, in general, infectious disease pandemics and flu as a virus that’s a very high risk of causing a pandemic are really something that we should be very concerned about.
Matt: So, if a flu pandemic is potentially a looming catastrophe, how do we protect ourselves?
Bethany: Well, there are some really straightforward things. Wash your hands thoroughly. Cover your cough. Some of Seema’s research is showing things like getting the air moving inside your house can help slow transmission of the flu. So even cracking a window every once in a while could help.
And, according to a recent study at the University of California, Davis, try not to speak loudly if you think you’re sick. Researchers found that the louder people spoke, the more airborne particles they emitted. So loudness is a potential factor in spreading airborne diseases.
Matt: So, like, don’t shout on people? Always good advice.
Bethany: Then, like we said early on, the CDC recommends getting the flu vaccine.
Matt: Question for you, Bethany. Did our experts get their flu shots?
Bethany: Let’s ask them.
Bethany (in interview): Do you get a flu shot every year?
Seema Lakdawala: I do, I do get a flu shot every year. And my kids get a flu shot every year. And my husband gets a flu shot every year.
Bethany: That was my next question.
[Laughter]
Bethany: Do you get a flu shot every year?
Adam Lauring: I do. I’m required to. [Laughter] I do think it’s a good idea in general. But, yes, so at the University of Michigan, and at most medical centers, its people are required to get a flu shot. But everyone in my family gets a flu shot too.
Bethany: Do you get a flu shot every year?
Jesse Bloom:I get a flu shot. Almost every year. I’m certainly a big believer in vaccines, and there are many vaccines, such as the MMR vaccine, the measles vaccine, things like that, which are just some the most phenomenal things that medical science has produced. I mean, I would say, if you’re not getting, for instance, the MMR vaccine or something like that, you’re crazy because we know these things have done a great job of protecting you against really bad diseases such as measles and mumps and things like that. I also get the flu vaccine most years, but as a flu researcher, I will acknowledge the flu vaccine is not as effective as those other vaccines. So if you get something like the measles vaccine, it reduces your risk of getting measles by more than 95%. An unfortunate part of the flu vaccine is it reduces your risk of getting flu, but only by maybe—it varies somewhat from year to year—let’s say only by 50%. So I do get the flu vaccine, but as a flu researcher, I recognize the flu vaccine isn’t as effective as some other vaccines, and I think a goal that I share with many other researchers in this field is that you’d like to make the flu vaccine function more like the measles vaccine.
Matt: So Bethany
Bethany: Yes, Matt.
Matt: I have a surprise for you.
Matt (at Kroger): All right. So. It is Jan. 9. I’m a little late to the party, but I am in the parking lot of a Kroger to get my flu shot. Let’s go inside.
Do you want to introduce yourself?
Megan Tabers: Sure. I’m Megan Tabers. I’m a registered pharmacist, a PharmD, and I’m going to give Matthew his flu shot today. And generally we do want to get patients in before the end of October is what is recommended by the CDC, but really any time between August to March is when you can get a flu shot. So it’s still good that you’re getting it, even though it’s a little bit later.
Matt: Nice.
Megan Tabers: So yeah. [Medical gear rustling] OK. And that was the flu shot.
Matt: Awesome. Well, thank you very much. That was very fast.
Megan Tabers: Yes.
Matt: I wish I had done it sooner.
Bethany (in studio): Good for you, Matt.
Matt: Thank you. So, if you haven’t already gotten it, get your flu vaccine, even if it’s not as effective as scientists would like.
Bethany: And remember, don’t cough, sneeze, or shout on other people; wash your hands frequently.
Matt: And if you do get the flu, stay home to recover, and open the window every once in a while.
Bethany: That’s right. And with any luck, you’ll be healthy when you hear the next episode of Stereo Chemistry in February.
Matt: Yes. Our next full episode is coming your way in just a few short weeks. We’ll be talking about climate change. Subscribe to Stereo Chemistry wherever you get your podcasts. And if you can’t find Stereo Chemistry wherever you get your podcasts, let us know. You can tweet at us using @cenmag or @MrMattDavenport, and we will get on that.
Advertisement
Stereo Chemistry is a production of C&EN, the newsmagazine of the American Chemical Society. This episode was written by Bethany Halford and produced by me. It was edited by Lauren Wolf and Amanda Yarnell. And our fantastic copyeditor is Sabrina Ashwell.
You heard several songs during this episode, and I’m going to tell you what they all were in chronological order. There was “So Easy,” by Stanley Gurvich, “Busy World,” by Lance Conrad, “Cold,” by Anthony Lazaro, “Lunas” by Tomas Novoa, and “On Air,” by Assaf Ayalon.
And that about does it for this episode.
Bethany: Thanks for listening.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter