Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Energy Storage
Microscopy method tracks lithium ions and dendrites in batteries
Study ties dendrite-growth mechanism to ion concentration near metal electrode
by Mitch Jacoby
August 23, 2018
| A version of this story appeared in
Volume 96, Issue 34

By borrowing a spectroscopy method often used to analyze biological specimens, researchers have developed a high-resolution microscopy technique that can monitor electrochemical processes occurring in batteries in real time. They used the method to probe quickly changing ion concentrations in an electrolyte solution as well as the mechanism of dendrite growth in lithium batteries.
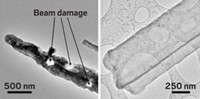
Li-ion batteries could hold about 10 times as much energy as today’s commercial varieties do, if the commonly used graphite anode were replaced with one made from lithium metal. But that replacement poses safety risks. During charging, lithium can accumulate on the metal anode in the form of dendrites. These spikey structures can grow, pierce the film that separates the positive and negative electrodes, and short-circuit the battery, possibly causing a fire or explosion.
To uncover the details of the mechanism behind dendrite growth, researchers have used electron microscopy and other imaging techniques on electrochemical cells. But important questions remain unanswered. For example, how does the concentration of Li+ ions in the electrolyte solution near the electrode affect dendrite growth?
At the American Chemical Society national meeting in Boston yesterday, Yuan Yang of Columbia University offered an answer to that question. Speaking at a symposium in the Division of Energy & Fuels, Yang described results of a study in which his team used stimulated Raman scattering (SRS) microscopy to track ions in a battery electrolyte solution and to simultaneously image growing lithium dendrites.
Yang explained that SRS is a two-laser-pulse technique that can enhance analytical signals by more than a factor of 10 million compared with a related, more common one-laser Raman spectroscopy method. With that improvement, the two-laser method was able to quickly generate three-dimensional images with high spatial resolution (~300 nm) and track ion transport at millimolar concentration levels.
The new study, which was conducted in a customized electrochemical cell, uncovered three stages of dendrite growth. When the concentration of Li+ ions in the layer of liquid immediately adjacent to the electrode is comparable to the concentration away from the electrode, lithium forms uniform, smooth moss-like deposits. When the ion concentration near the electrode is partially depleted, the metal accumulates in a mixed mode, with mossy patches and some dendrites. In contrast, high levels of Li+ ion depletion lead to fast growth of dendrites (Nat. Commun. 2018, DOI: 10.1038/s41467-018-05289-z). Ion concentration at the electrode surface is affected by various factors including electrode geometry and charging conditions.
“The clever use of SRS microscopy to visualize the electrolyte concentration in an operating electrochemical cell is a real imaging breakthrough,” said Martin Bazant, a specialist in ion-transport phenomena at Massachusetts Institute of Technology. These results have implications for the design of safe rechargeable metal batteries, he added. Indeed, the Columbia team found that adding a nanometer-thin layer of lithium phosphate to the electrode homogenized the ion concentration, preventing severe ion depletion and dendrite growth.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter