Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Nuclear Power
Proposed nuclear waste storage materials may have a corrosion problem
Current safety assessment models don’t take into account the synergistic reactions between different materials used in these storage systems
by Prachi Patel, special to C&EN
January 30, 2020
| A version of this story appeared in
Volume 98, Issue 5

Many countries including the US plan to store nuclear waste in underground repositories. But scientists might not have a complete picture of the safety of the storage materials involved in these plans, according to a new study. The report demonstrates that interactions between the different materials used for these storage systems could accelerate their corrosion when they are exposed to water, increasing the chance of the radioactive waste leaking into the environment (Nat. Mater. 2020, DOI: 10.1038/s41563-019-0579-x).
Spent fuel from nuclear power reactors can remain highly radioactive for thousands of years. The waste is typically kept in large ponds or concrete vaults at reactor sites to cool down. But for permanent storage, several countries plan to turn the waste into glass or ceramic, and encase the solids in stainless steel canisters to store in repositories.
Each of these materials separately resist corrosion in tests that simulate the cold, wet conditions that could exist in repositories, says Xiaolei Guo, a materials scientist and corrosion researcher at Ohio State University. But current nuclear waste storage assessment models do not consider the chemical interactions between these materials when they are brought together. According to experiments by Guo and his colleagues, if water gets inside the steel canister and works its way into the tiny gap between the canister and the glass or ceramic, it can trigger a string of corrosive reactions that degrades both materials.
The elemental soup found in these materials is the culprit. The glass is made of oxides of silicon, aluminum, calcium, and other elements, while the ceramic is an oxide of barium, chromium, and aluminum. And stainless steel has mainly iron with some nickel and chromium. Synergistic chemical interactions between these elements causes the corrosion, Guo says.

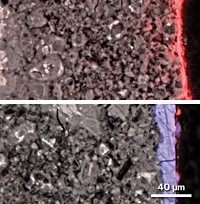
The researchers observed these reactions when they pressed a piece of nuclear waste glass or ceramic—minus radioactive elements—to a stainless steel plate and immersed them in a sodium chloride solution for 30 days.
At the end of the experiment, the steel was pitted along the edge where it touched the glass or ceramic. For the ceramic, this corrosion started after just 7 days and was much worse than what was observed with the glass at the end of the 30 days. Imaging and chemical analysis revealed a gel layer on the glass around the edges that contained mainly iron and silicon with a mix of other elements. Further away from the edges tiny crystalline particles made of silicon-rich minerals dotted the glass.
Guo explains that metal ions from the steel first dissolve into the water and react with water, releasing protons that increase the local acidity. This acidity corrodes the steel and the glass or ceramic, releasing ions that can further enhance the corrosion of the steel, creating a feedback loop.
Electrons released during these reactions reduce oxygen and create an alkaline environment that causes silicon to leach out of the glass to form the observed crystalline particles. The corrosion is worse with the ceramic because of the highly reactive chromium ions released from both the steel and the ceramic.
Researchers have known for some time that concrete found in nuclear waste repositories can influence the corrosion of nuclear waste glass, says Mathieu Bauchy, a civil and environmental engineer at the University of California, Los Angeles. “However, the fact that stainless steel could have such a sudden and severe detrimental effect on the corrosion of glasses and ceramic phases is rather unexpected.”
These results could impact the projected service life of nuclear waste in repositories. “Present models may be underestimating the risk of corrosion in nuclear waste disposal systems,” Bauchy says. However, he points out that the study is limited to 30 days and the mechanisms observed should be tested over more extended time periods. As of press time, the US Department of Energy, which oversees plans for storing nuclear waste, had no comment about the study.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter