Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
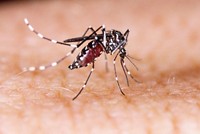
Pesticides
US EPA green-lights genetically modified mosquito
by Britt E. Erickson
May 8, 2020
| A version of this story appeared in
Volume 98, Issue 18

The US Environmental Protection Agency (EPA) has granted UK-based Oxitec an experimental use permit to test whether releasing genetically modified Aedes aegypti mosquitoes can reduce the population of the pest, which transmits diseases such as Zika, chikungunya, and dengue. The company plans to release male mosquitoes that have been genetically modified to produce a protein that prevents their female offspring from surviving. The experiment will be conducted for 2 years in Florida beginning this summer and in Texas starting next year. Oxitec has completed similar tests in Brazil that the company says successfully suppressed the wild Aedes aegypti population. “Our aim is to empower governments and communities of all sizes to effectively and sustainably control these disease-spreading mosquitoes without harmful impact on the environment and without complex, costly operations,” Oxitec CEO Grey Frandsen says in a statement. The US Food and Drug Administration concluded in 2016 that Oxitec’s genetically modified mosquito will not have significant impacts on the environment. In 2017, the FDA transferred regulatory jurisdiction for the technology to the EPA. Federal permit in hand, Oxitec is now seeking approval from state and local regulators.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter