Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Coatings
New device could reduce toxic chemotherapy side effects
Chemically coated, 3-D printed cylinder removes a common chemotherapy drug from the bloodstream
by Emma Hiolski, special to C&EN
January 14, 2019

A device smaller than a AAA battery may have the potential to shield healthy tissue from the devastating side effects of chemotherapy. The customizable, 3-D-printed device is placed in a blood vessel downstream of a target organ and can absorb a common chemotherapy drug directly from the bloodstream before it spreads to the rest of the body (ACS Cent. Sci. 2019, DOI: 10.1021/acscentsci.8b00700).
Steven W. Hetts of the University of California, San Francisco, came up with the idea for drug recapture about eight years ago. Part of Hetts’s work as a neuroradiologist involves threading catheters through a patient’s blood vessels to deliver chemotherapy drugs directly to a tumor or particular organ in a procedure called intra-arterial chemotherapy. Some 50 to 80% of the injected drug passes through the target, however, and enters the rest of the body.
One common chemotherapy drug, doxorubicin, is highly effective at treating various cancers, but cumulative exposure from repeated treatments can lead to heart failure. Hetts wondered if it was possible to capture doxorubicin as it exited a target organ—in this case the liver—before it spread throughout the body. Currently, liver cancer treatment involves regular intra-arterial chemotherapy to keep tumors in check until a liver transplant can happen, Hetts says. A device that could recapture doxorubicin on its way out of the liver would not only lower the risk of heart failure but also reduce other side effects such as hair loss and immune suppression. It might even allow the delivery of higher doxorubicin doses to the liver for more effective treatment, he says.
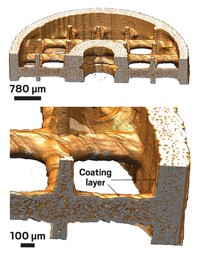
To further develop this idea, Hetts turned to collaborators at the University of California, Berkeley. Led by chemical engineer Nitash P. Balsara and postdoctoral researcher Hee Jeung Oh, the team envisioned a porous device with a high surface area to maximize contact with the bloodstream. They designed a square lattice structure within a 5-mm-wide, 30-mm-long cylinder that could be placed into a patient’s vein during intra-arterial chemotherapy via the same minimally invasive surgery used to deliver the doxorubicin. The device would need to stay in place for only two hours during this treatment.
The device was 3-D printed by Balsara’s longtime collaborator, Joseph M. DeSimone, CEO and cofounder of the tech company Carbon, Inc. The company’s light-activated version of 3-D printing provides fast and accurate production; it takes minutes to print a single device, DeSimone says.
The device is made of poly(ethylene glycol) diacrylate, a biocompatible and foul-resistant material. Balsara’s team then coated this scaffold with a polystyrenesulfonate-containing block copolymer that sticks to the scaffold and irreversibly binds doxorubicin.
To confirm that the device could indeed absorb doxorubicin from the bloodstream, the team tested it in pigs, inserting two absorbers side by side into a vein of similar diameter to the vein leaving the human liver, where they might one day be deployed. The researchers injected a clinically relevant dose of doxorubicin through a catheter just upstream of the devices, and measured drug concentration both upstream and downstream of the devices. The absorbers captured an average of 64% of the administered doxorubicin and caused no adverse effects to the blood vessels during the 90-minute experiments.
Advertisement
Though the team still wants to optimize the chemical binding properties of the device, they are excited by its potential. “I think this is a realistic device and approach to reducing the toxicity of chemotherapy and will help a large number of patients,” Hetts says. Using magnetic resonance imaging or computed tomography scans to create 3-D images of an individual’s blood vessels could eventually allow the absorber to be custom printed to fit that person’s vein, he adds.
Sharon C. Glotzer, of the University of Michigan, calls the study “another great application of fundamental chemical engineering to problems in human health.” Glotzer also highlights how the approach could be applied to other medical situations in which toxins are unwanted. Targeting other toxins, however, would require developing copolymers that bind other molecules. “I see this work as a first step, but a very big first step,” she says.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter