Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Inorganic Chemistry
Chemistry In Pictures
Chemistry in Pictures: Violets are blue, and roses are black
by Manny Morone
February 11, 2020

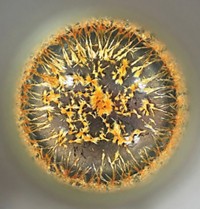
This cluster of crystals wasn’t made with a mold, but rather Bhakti Patel made it in a lab. She’s a PhD student in Joe Kolis’s lab at Clemson University, which works on developing new magnetic and nonlinear optical materials. Patel mixed the compounds V2O5, SnO2, and CsF using a high-pressure and high-temperature hydrothermal method, which mimics the way many mineral crystals form in nature. She hoped to make a new compound containing vanadium and tin in a single structure but instead ended up with two different compounds: “leaves” of Cs2SnF6, which are green due to V4+ impurities, and “petals” of Cs2V3O8, which are black because of mixed V4+ and V5+ ions in the compound’s lattice.
Submitted by Bhakti Patel. You can follow her on Instagram at @crystalcrazeblue.
Do science. Take pictures. Win money. Enter our photo contest here.
Related C&EN Content:




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter