Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Metal-Organic Frameworks
Hydrogen and methane-adsorbing MOF boasts impressive combination of properties
Aluminum-based sorbent with exceptional gas-storage properties may give clean-burning fuels a transportation boost
by Mitch Jacoby
April 20, 2020

A new solid sorbent could pave the way toward inexpensive and safe storage of hydrogen and methane, which could serve as environmentally friendlier alternatives to conventional liquid transportation fuels. The material balances many of the properties that researchers have been looking for in gas sorbents (Science 2020, DOI: 10.1126/science.aaz8881).
Replacing gasoline and diesel fuel with clean-burning hydrogen, which can power zero-emission fuel-cell vehicles, or with methane, which generates relatively low levels of combustion products, could go a long way toward reducing atmospheric levels of carbon dioxide, a greenhouse gas. One challenge impeding broad implementation of such low-emission vehicles is coming up with a low-cost on-board fuel system that can store enough gas at moderate pressure to enable practical driving distances between fill-ups. In principle, gas-adsorbing solids such as metal-organic frameworks, or MOFs, could help in this quest because ordinary fuel tanks containing these sponge-like materials could hold more gas at low pressure than costly specialized tanks can hold at high pressure.
MOFs are a large family of porous crystalline solids composed of metal ions or clusters joined by organic linkers. By tailoring the molecular building blocks, which control internal pore sizes and other features, researchers have previously made MOFs with extremely high internal surface areas. Some MOFs adsorb exceptional amounts of gases such as hydrogen on a per-weight basis. Others work impressively only on a per-volume basis. Still others soak up large amounts of several gases but don’t easily release them.
A team led by Zhijie Chen, Penghao Li, and Omar K. Farha of Northwestern University set out to balance these properties in a single MOF by combining various computational and experimental methods. For example, the team evaluated thousands of MOFs to explore tradeoffs between properties such as internal empty space—or void fraction—largest pore diameter, and the strength of host–guest interactions.
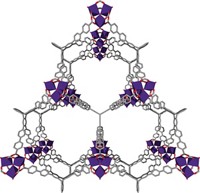
The study led them to NU-1501-Al, which is composed of trinuclear aluminum centers and triptycene-like organic ligands (the researchers also made an iron analog). The new MOF exhibits surface-area and reversible, or deliverable, gas-uptake values that, taken individually, are outstanding but not record setting. The combination of these properties in a single material, however, is unprecedented.
The Al-based MOF has a surface area of 7,310 m2 per gram (one of the highest) and 2,060 m2 per cm3. It boasts a methane uptake value of 0.66 g per gram of MOF, which exceeds the Department of Energy’s gravimetric target value (0.5 g per g), and it nearly meets the DOE’s volumetric target, 263 cm3per cm3. The new material also exhibits one of the highest capacities for deliverable hydrogen both on a weight and volume basis—roughly 14% by weight and 46 g per L.
“This is a spectacular demonstration of how being able to control matter on the atomic, molecular, and framework levels in one extended chemical structure, leads to amazing properties not achievable without such precise control,” says MOF pioneer Omar M. Yaghi of the University of California, Berkeley.
Seth M. Cohen of the University of California, San Diego, another MOF specialist, remarks that through rigorous computational analysis, the team simultaneously maximized volumetric and gravimetric capacities in a single compound. “The combination of computation and experiment has resulted in impressive properties in the MOF.”
CORRECTION
This story was updated on April 23, 2020, to correct "absorbing" to "adsorbing" in the headline.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter