Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Polymers
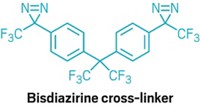
Double diazirine cross-links virtually any organic polymer
Molecule also acts as a universal adhesive for polymers, thanks to functional groups that form carbenes
by Bethany Halford
November 14, 2019
| A version of this story appeared in
Volume 97, Issue 45
By tethering two reactive diazirine groups together, chemists have created a compound that cross-links essentially any organic polymer, potentially improving properties like mechanical strength and corrosion resistance. The new cross-linking molecule also functions as a universal adhesive for polymers, making it possible to stick virtually any polymer to itself or a different material.
Cross-linking polymers simply means adding bonds across polymer strands, explains Jeremy E. Wulff, a chemistry professor at the University of Victoria who led the work. Silicone bakeware, certain types of joint replacements, and many modern plumbing lines are made from cross-linked polymers.
“There are plenty of methods available for cross-linking polymers, but each of them is custom designed for the particular polymer,” Wulff says. “What we tried to do was to come up with a way that we could cross-link pretty much any polymer using the same set of conditions.”
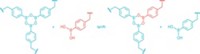
To do so, Wulff’s team took a cue from chemical biologists, who have been using diazirines—three-membered rings with one carbon and two double-bonded nitrogens—for decades to link small molecules to proteins. When exposed to light or heat, diazirines break down into carbenes, which promiscuously insert into C–H, N–H, and O–H bonds. By creating a molecule with a diazirine at each end, Wulff and coworkers reasoned, they could create a general cross-linker (Science 2019, DOI: 10.1126/science.aay6230).
Using just a small amount of the bisdiazirine, the chemists were able to cross-link several types of polymers, including polypropylene, which resists cross-linking. “We see this cross-linker as a magic pixie dust, where we can take an existing polymer material and just sprinkle in a little bit of the cross-linker and change the material’s properties,” Wulff says.
The cross-linker also works as a universal adhesive for polymers, allowing the chemists to glue pieces of notoriously slippery materials like polyethylene together or to other plastics or metals.
Karen L. Wooley, a polymer expert at Texas A&M University, says the cross-linker could add value to polyolefins, which can be tough to recycle because of their chemical inertness. “The reported bisdiazirine-generated carbene chemistry could be useful for cross-linking-based upcycling of polyolefin properties while also serving as a strategy for further chemical modification for polyolefins and other polymer materials as well,” she says.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter