Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Polymers
Reversible polymer chemistry could make a truly recyclable plastic
Polymer forms through click chemistry reactions and then reverts back to monomer building blocks in strong acid
by Sam Lemonick
May 1, 2019
| A version of this story appeared in
Volume 97, Issue 18

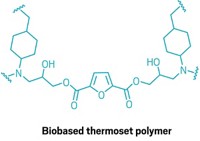
When recycling plastics, contamination and chemistry conspire to degrade the quality of each recycled generation. In a perfect world, a polymer could be converted to its monomer building blocks, then made into new plastic over and over. Researchers have been working to develop such chemistry, and Brett Helms and colleagues from Lawrence Berkeley National Laboratory now demonstrate a new approach (Nat. Chem. 2019, DOI: 10.1038/s41557-019-0249-2).
Their polymer is based on a network of reversible diketoenamine bonds formed in a ball mill using click chemistry reactions between triketones and polyamines like tris(2-aminoethyl)amine. Helms says that the resulting plastics, which the group refers to as poly(diketoenamine)s (PDKs), have mechanical properties on par with thermoplastic polyurethanes, which are used in products such as mobile phone cases and shoe soles. PDKs are in a class of plastics called vitrimers that are characterized by a crosslinked polymer network. The materials, which behave like glasses, have been explored as recyclable plastics by other chemists.
To convert the PDKs back to their monomer building blocks, the group uses 5 M sulfuric acid. The researchers could recover more than 90% of the triketone and polyamine monomers after 12 h in the acid. They could then separate the monomers from additives and other polymers left in the acid solution. For example, they could dissolve and extract the triketones and amines from dyed plastics, mixtures of different plastic types, and plastics mixed with flame retardants.
This research shows more of what vitrimers can do, says Eric Drockenmuller, a polymer chemist at Claude Bernard University Lyon 1. “Besides showing properties typical of the best vitrimers developed so far,” he says, Helms’ group also demonstrated a new approach to so-called closed-loop recycling.
But Jinwen Zhang, who studies vitrimers at Washington State University, points out using a strong acid comes with its own environmental concerns in terms of waste disposal. He also says for PDKs to be commercially practical, they will need a cheap feedstock. Helms says that his team has made renewable feedstocks a priority from the start.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter