Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Pharmaceuticals
Outsourcing tales from the drug services industry trenches
Small drug companies often don’t have the resources to make their own products. Here are stories of how they work with service firms that handle the production for them
by Michael McCoy and Rick Mullin
June 24, 2019
| A version of this story appeared in
Volume 97, Issue 25

Credit: Yang H. Ku/C&EN/Shutterstock
COVER STORY
A biotech takes a risk on a distant supplier
Lexicon hires the Chinese service firm WuXi AppTec and is pleasantly surprised
by Michael McCoy

In 2004, Winston Wu was at a crossroads. A principal scientist and project leader at Schering-Plough, he had worked as a chemist at big companies for almost 12 years. He had learned a lot about chemical development but was finding his opportunities for growth to be limited.
He considered joining a contract research or manufacturing firm, possibly one in his native China. Instead, he accepted a job at a small biotech called Lexicon Pharmaceuticals. The firm was about to advance a compound into development, and Wu reasoned that he could aid that effort with the skills he had developed at Schering-Plough.
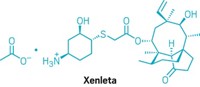
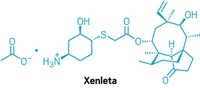
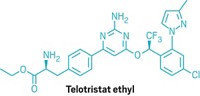
Xermelo
Active ingredient: Telotristat ethyl
Discovered: 2006 by Lexicon Pharmaceuticals scientists
Indication: Diarrhea associated with carcinoid tumors
2018 sales: $25 million

That is indeed how things worked out for Wu, who today is Lexicon’s vice president of chemical development. In early 2017, Lexicon won US Food and Drug Administration approval for Xermelo, a treatment for diarrhea that can accompany a rare cancer called a carcinoid tumor. Through his management of outsourcing partnerships with companies, including the Chinese pharmaceutical services firm WuXi AppTec, Wu played a big role in getting Xermelo approved.
The active pharmaceutical ingredient (API) in Xermelo is telotristat ethyl, a prodrug of telotristat. Telotristat inhibits tryptophan hydroxylase, an enzyme involved in the body’s synthesis of serotonin. People with carcinoid tumors overproduce serotonin, which plays a role in regulating the gastrointestinal tract. By inhibiting tryptophan hydroxylase, Xermelo reduces serotonin production and thus the frequency of the diarrhea that accompanies the disease.
Telotristat ethyl is one of three related molecules discovered by Lexicon scientists in 2006. Wu explains that all three molecules share a phenylalanine group on the left and an aminopyrimidine group in the middle; what makes Xermelo unique is the group on the right, which comes from a chiral alcohol.
When Wu joined Lexicon in 2005, he was put in charge of process chemistry. At the time, he says, the pharmaceutical chemistry firm that Lexicon was working with was unable to scale up the synthesis of a boronic acid needed to connect the phenylalanine group to the aminopyrimidine. Wu decided to give WuXi a try.
It was a bit of a radical move. Today, WuXi says it works on more than 50 APIs for drugs that are either commercial or in Phase III clinical trials. But at the time, the Chinese firm was only about 5 years old. Although it was growing quickly in size and reputation, its main customers were large drug companies with deep experience in managing outsourcing partners.
Wu, though, knew WuXi from his Schering-Plough days. In fact, the Chinese firm had successfully scaled up a compound for which Wu had developed a process. And two executives at WuXi’s STA Pharmaceutical drug manufacturing division came from Schering-Plough: Minzhang Chen, who today is STA’s CEO, and Xiaoyong Fu, its senior vice president for API development and commercialization.
“Schering was pretty happy with WuXi,” Wu says, “so when I needed services at Lexicon, they came to mind.”
Rather than keep working with the boronic acid, Lexicon developed a process for a chemically equivalent boronic ester, but it had difficulty with the isolation step. Wu sent a technical package to WuXi, which was able to solve the problem. The ester was prone to hydrolyzing into the acid, complicating the crystallization. Chemists at WuXi’s labs in Shanghai’s Waigaoqiao district stopped the hydrolyzing with an ingredient that transformed the acid in situ back to the ester. The firm went on to make metric-ton quantities of the ester for all three members of the compound family at its facility in the Shanghai Chemical Industry Park.
WuXi’s success with the boronic ester convinced Lexicon’s top brass of its skills, and Wu sent more projects its way. One was a Suzuki coupling to connect the phenylalanine and aminopyrimidine pieces into an intermediate for the three APIs.
At first, Lexicon sent the intermediate to a Western pharmaceutical services firm for conversion into telotristat ethyl, but by 2010 WuXi was also making APIs for Lexicon. Wu says the company was particularly helpful during a difficult period in developing the drug.
The US FDA had signed off on Lexicon’s data package for the choice of regulatory starting materials (RSMs)—defined as the first intermediates in a multistep synthesis to be made under current good manufacturing practice (cGMP) conditions. But the European Medicines Agency (EMA) had different requirements. While the FDA was satisfied with designating the chiral alcohol and the phenylalanine-aminopyrimidine intermediates as RSMs, the Europeans wanted more steps to be conducted under cGMP.
“That was a period when I was glad to have WuXi,” Wu recalls. “They had so many chemists and instrumentation and could react quickly when we needed a lot of data generated in a short period of time.”
Youchu Wang, a WuXi STA vice president who leads the firm’s collaboration with Lexicon, remembers that time as well. “The RSM pushback by EMA caused a significant amount of work,” Wang says. In addition to more steps under cGMP, EMA required impurity profiling and additional quality risk assessments. WuXi was able to complete the work on time and without affecting the EMA filing, he says.
Beyond those changes, Wang says, WuXi made a number of synthesis and process improvements to four reaction steps. For example, the original process had a late esterification step that gave a final intermediate a complicated impurity profile. By investigating the genesis, fate, and purge of impurities, WuXi was able to make the process more robust, Wang says. Additionally, by understanding reaction kinetics, the firm was able to reduce the solvent volume to one-third of its original and decrease the esterification time significantly, he says.
Efforts like these beyond what was required explain why Wu is impressed with the Chinese company. The conventional wisdom says Western chemistry service providers are closer and more communicative partners for biotech firms than are their Asian competitors. Wu reports otherwise.
“We found WuXi to be more flexible in its business model and more collaborative,” he says. For example, it showed a willingness to make changes to its service agreement without an onerous “change of scope” modification to the contract. “I spent less time dealing with contracts,” Wu says.
Xermelo is an orphan drug that the FDA approved for a narrow indication: use with somatostatin analog (SSA) therapy to treat adults with carcinoid tumor–related diarrhea that is not adequately controlled by SSA therapy alone. As Lexicon reported in March, Xermelo’s sales in 2018, its first full year on the market in the US and Europe, were a modest $25 million.
But Lexicon is exploring other potential uses for Xermelo because preclinical data suggest that telotristat ethyl’s ability to inhibit serotonin synthesis may also reduce cancer cell growth in several cancers known collectively as biliary tract cancers. The firm is in the midst of a Phase II study of Xermelo in this application.
With additional demand for telotristat ethyl a possibility, WuXi has been working to scale up a new synthesis that Wu’s group developed for one of the drug’s intermediates. The new synthesis is more efficient, is cleaner, and generates fewer impurities, Wu says, but it was plagued by one particular impurity that proved hard to eliminate.
“WuXi came up with an ingenious method to remove it,” Wu says. “I was completely surprised.”
Given WuXi’s scientific prowess and its proactive approach, it’s no wonder that Wu keeps going back to the Chinese firm, including for other molecules in Lexicon’s pipeline. “WuXi is one of our most preferred suppliers for APIs,” he says.
Ticking a new box in enzyme chemistry
Going beyond its comfort zone in biocatalysis, Almac immobilizes an enzyme and manufactures a polymer for Alcresta’s lipase cartridge device
by Rick Mullin

With their first start-up, Alnara Pharmaceuticals, biotech entrepreneurs Alexey Margolin and Robert Gallotto developed a pancreatic enzyme replacement therapy, liprotamase, to treat exocrine pancreatic insufficiency—an inability to break down fats experienced by people with cystic fibrosis and other diseases. The product, which includes a microbially derived lipase enzyme, replaced a lipase that for nearly 100 years had been derived from pig pancreases.
After Eli Lilly and Company acquired Alnara in 2010, the entrepreneurs had a new idea: address concerns that ingesting pancreatic enzyme replacements can cause bowel obstruction and inflammation.
Relizorb
Generic name: Immobilized lipase cartridge
Commercial launch: 2016
Innovation: Feeding-tube device eliminating ingestion of lipase-replacement therapies
Components: Immobilized lipase enzyme bonded to methacrylate polymer beads

Their new venture, Alcresta Therapeutics, launched in 2011 to develop a device that breaks down fats externally by connecting to a feeding tube. The result was Relizorb, a cartridge launched in 2016 that employs a lipase enzyme immobilized on polymer beads. Triglycerides in the feeding formula pass through the device and break down to fatty acids and monoglycerides before ingestion, with none of the enzyme leaving the cartridge.
Convinced from bench experiments that the device would work, the partners prepared to scale up for clinical and commercial production. They secured food-grade lipase from a Japanese supplier but needed to immobilize the enzyme on a polymer bead they’d designed. David Brown, vice president of technical operations at Alcresta, whose task was to find a contract services firm, found that enzyme immobilization is a peculiar niche expertise.
Brown, who worked at Alnara and was involved in developing the fermentation process for liprotamase, was familiar with the contract development and manufacturing organization (CDMO) market by the time he rejoined Margolin and Gallotto at Alcresta. “When Alnara was sold to Lilly, I went to Patheon,” he says. There, he immersed himself in the world of finished drug development and manufacturing services.
Alcresta placed him in new territory. “It was really kind of an exciting time for me because I had just come out of this classical CDMO world, and I was following the trends of what big pharma needed from CDMOs,” he says. “The whole world of enzymes, and enzyme immobilization in particular, was much smaller and not necessarily of interest to the big firm I represented.”
After exploring the market, Brown identified 28 service providers with enzyme-immobilization expertise, but only 4 of them also had experience with suspension polymerization—the process for synthesizing the bead. The company Alcresta eventually signed with, Almac Sciences, wasn’t on the short list.
“Companies with experience in both immobilization and suspension polymerization recognized they were in a unique position and were larger companies,” Brown says. “Frankly, they wanted to make a bigger deal of it than Alcresta could stomach at the time.”
As he widened his search he decided to take another look at Almac. Brown saw that the Northern Irish firm’s biocatalysis group had expertise in using enzymes as catalysts and might be ready for a new challenge: “They had never made a product for a product’s sake.”
According to Tom Moody, vice president of technology development and commercialization at Almac, the biocatalysis division was primed for the job.
“They were looking for someone with analytical chemistry around enzyme transformation, someone who understood enzyme production and purification, someone who knew about how to take R&D projects that involve chemistry and biochemistry right through to production,” he says.
“They wanted someone who can immobilize the enzymes, who understands heterogeneous catalysis, someone who could do design-of-experiment studies for any robust process that can be scaled commercially, and someone who can look at a second-generation product, which we are currently doing. And they wanted someone who can produce at commercial scale and deliver multiton of product,” he continues. “We ticked all the boxes.”
The commercial production box was ticked, Moody says, thanks to Almac Group’s 2015 acquisition of Arran Chemical. Although Almac manufactures to the US Food and Drug Administration’s current good manufacturing practice (cGMP) quality standard at its headquarters in Northern Ireland, it operates Arran as a non-cGMP site for the production of drug intermediates and nonpharmaceutical chemicals. The work for Alcresta is done at Arran to the ISO 9001 quality standard.
And it requires a lot of chemistry, Moody points out. “A lot of people hear biocatalysis, and they think it’s a black box,” he says. “Fundamentally, it’s synthetic organic chemistry. Given the way we formulate some of these processes, rather than call them biocatalysts, we call them catalysts.”
Almac went to work quickly on the box it could not tick—suspension polymerization. “It was quite a steep learning curve,” recalls Scott Wharry, senior research and manufacturing team leader in the company’s biocatalysis group, noting that Alcresta wanted the enzyme-immobilization provider to be a key supplier of the beads.
“There was lots of background reading and speaking to various people with experience and just planning a systematic approach to investigate the various components of the reaction,” Wharry says.
Engineers at Almac assisted in developing a model that could be scaled up for commercial production. “We ended up designing and installing a reactor specifically for that,” Wharry says. “After developing the process and making appropriately sized beads at laboratory scale, we increased in scale until we were manufacturing at approximately 1,000 L scale.” Although the beads are now made by another firm that is not involved in enzyme immobilization, Almac remains ready as a supplier.
Ensuring product purity is a key challenge, according to Wharry. “We did an extensive bit of analytical work to help Alcresta with the regulatory bodies to ensure no residual components of the bead formation or residual protein would wash off the beads and into the patient.”
The immobilization is a standard covalent bonding reaction, Wharry says, but there are some nuances. “The immobilization is a tricky process because you have to get to a certain level of protein attachment,” he says. “You need to make sure there is no leachable protein or nonbonding protein or other impurity.” Nor can there be microbial growth on the beads, he says.
“There are certain functional groups on the bead that allow the enzyme we are grafting to retain high activity,” Wharry continues. “There is an epoxide on the bead, and you have amine functionality on the protein. We are covalently binding the enzyme to the surface.”
“Once it’s grafted,” Moody adds, “it’s grafted.”
Brown concurs that the chemistry is tricky. “This is wet chemistry in tanks, yet it’s two phases, solid and liquid, with some flammable solvents,” he says. “And the beads are somewhat fragile and require delicate handling.” At 0.5 mm across, the methacrylate polymer beads are larger than the polymers that are usually produced through suspension polymerization.
“We have an effective surface area of 250 m2/g, which is about the size of a tennis court,” Brown says. “The beads that got us to clinical trials were an incredible achievement.”
So, he suggests, was finding the right service firm to connect the enzyme to the bead. Brown says he valued Almac’s willingness to develop an immobilized enzyme as a product rather than as a catalyst and its willingness to take on a unique challenge. Beyond its chemistry expertise, Brown adds, the quality that most sold Alcresta on Almac can be summed up in one word: attitude.
Behind the curtain
A look at 3 antibiotic manufacturing contracts reveals committed partners but a tough market
by
Michael McCoy and Rick Mullin
The contract drug manufacturing industry is something of a black box. Contracts between pharmaceutical firms and their active pharmaceutical ingredient (API) suppliers are almost always confidential. But a sliver of light comes through in the annual 10-K reports and other filings that publicly traded drug companies must submit to the US Securities and Exchange Commission. In these reports, some, though not all, small biotech firms disclose their manufacturing partners. By scouring the 10-Ks of biotechs that won new drug approvals in 2018, C&EN uncovered the following three contract manufacturing relationships involving new antibiotics. One thing these stories make clear: developing a new antibiotic is hard; making money from it is even harder.
Paratek Pharmaceuticals

Paratek Pharmaceuticals won US Food and Drug Administration approval for Nuzyra (omadacycline) in October 2018. The drug, a modernized tetracycline, treats community-acquired bacterial pneumonia and acute skin and skin-structure infections. To manufacture Nuzyra, which launched commercially in February, Boston-based Paratek assembled a veritable village of European contract manufacturing partners.

Omadacycline is produced in a three-step synthesis starting with minocycline, a well-known tetracycline antibiotic.
In November 2016, Paratek contracted with the Portuguese antibiotics maker Cipan to produce both minocycline and crude omadacycline. Soon after the FDA approved Nuzyra, Cipan announced plans to invest close to $7 million to expand its facility in Vala do Carregado, Portugal, to support the contract with Paratek. Cipan says it is one of only a few companies in the world to use fermentation and chemical synthesis together to produce pharmaceuticals.
Paratek transfers crude omadacycline from Cipan to the Swiss API contractor Carbogen Amcis, which upgrades the crude material into a high-purity form suitable for formulation into the finished drug. Paratek says it pays Carbogen in the “seven-digit US-dollar range” per batch it purchases.
The high-purity omadacycline then goes to Almac Pharma Services, a Northern Ireland–based company, for formulation into solid-dosage tablets. Omadacycline also goes to Patheon, part of Thermo Fisher Scientific, for packaging in injectable form at a facility in the UK.
In October, Paratek pointed to the approval of Nuzyra as an important milestone in light of ever-increasing antibiotic resistance, especially in the case of bacteria that cause pneumonia and skin infections. But initial signs are that uptake by the hospital community will be slow. In May, Paratek announced that Nuzyra’s sales in its first 8 weeks on the market were $1.3 million, a figure with which the firm said it was pleased.
And Paratek’s scientific team was honored in April with the American Chemical Society’s 2019 Heroes of Chemistry Award for its work on Nuzyra and a second tetracycline antibiotic, Seysara (sarecycline), that the FDA also approved last year. The Heroes of Chemistry Awards ceremony will take place during the fall ACS meeting in San Diego. ACS publishes C&EN.—MICHAEL MCCOY
Tetraphase Pharmaceuticals

Tetraphase Pharmaceuticals was founded in 2006 to commercialize a chemistry platform that emerged from the lab of Harvard University chemistry professor Andrew G. Myers.
As C&EN reported in 2010, Myers devised a fully synthetic route to tetracycline derivatives that enables chemists to make thousands of compounds with modifications at virtually any position on the molecule. Previously, tetracycline derivatives had been made through a semisynthetic route that enabled modifications only at the C7 and C9 positions.

One of the compounds to emerge from the technology at Tetraphase was the tetracycline eravacycline, and in August 2018 the FDA approved it for the treatment of complicated intra-abdominal infections. The firm launched eravacycline 2 months later under the brand name Xerava.
Like many small biotech firms, Tetraphase has no manufacturing facilities of its own. “We do not own or operate manufacturing facilities for the production of Xerava or any of our product candidates, nor do we have plans to develop our own manufacturing operations in the foreseeable future,” the company’s most recent 10-K report says.
Instead, Tetraphase outsources. In 2014, it signed an agreement under which the French pharmaceutical services firm Novasep started producing clinical supplies of eravacycline. In October 2017, the pair extended the deal to include commercial-scale manufacturing at Novasep’s facility in Chasse-sur-Rhône, France.
Tetraphase transfers the API produced by Novasep to Patheon, part of Thermo Fisher Scientific, for formulation into a finished injectable liquid. Patheon carries out the step at facilities in Greenville, North Carolina, and Ferentino, Italy.
Xerava sales are building slowly, reaching only $341,000 in the first quarter of 2019. But Tetraphase CEO Guy Macdonald is optimistic, telling investors that interest is growing among physicians in US hospitals and health-care institutions. Xerava has been added to more than 200 formularies at “top-prescribing hospitals,” he said.—MICHAEL MCCOY
Achaogen

Achaogen, founded in 2002, entered the antibiotic fray with plans to target gram-negative bacteria with both antibodies and small molecules. The firm eventually settled on plazomicin, a small molecule that is designed to overcome resistance mechanisms affecting this class of bacteria, as its lead drug development program. Achaogen’s efforts yielded Zemdri, a treatment for adults with complicated urinary tract infections that the FDA approved in June 2018.

The treatment, developed in a program funded in part by the US Biomedical Advanced Research and Development Authority for up to $104 million, proved highly effective, registering a significant reduction in relapse of clinical symptoms of the infection after 28 days compared with the standard of care.
However, the advisory committee that voted to approve the drug to treat urinary tract infections declined to approve it to treat bloodstream infections. Hitting the market with approval for one narrow indication, Zemdri logged sales of only $800,000 last year.
In November, the company announced steps to reduce its operating expenses by 35–40% along with plans to evaluate its strategic alternatives. In April, it filed for bankruptcy and began the process of auctioning off its assets.
Back when its prospects looked brighter, Achaogen went looking for service providers to manufacture Zemdri. In 2017, it entered an agreement with the Portuguese pharmaceutical services firm Hovione for scale-up and production of plazomicin. Achaogen committed an initial $14.8 million to a 7-year contract with a potential value of $26.4 million.
Under the contract, Achaogen agreed to purchase at least 80% of its required quantity of plazomicin from Hovione for the first 3 years after FDA approval, after which it committed to buying 40–60%. Separately, it signed up Pfizer CentreOne to produce the finished intravenous form of the drug.
Achaogen announced earlier this month that it has agreed to sell its assets for approximately $16 million, plus potential royalties, to several buyers, including the Indian drugmaker Cipla, which will acquire worldwide rights excluding China to Zemdri. Qilu Antibiotics Pharmaceutical will acquire rights to the drug in China.
Achaogen’s liquidation comes as Zemdri is under review by the European Medicines Agency. The company’s second antibacterial candidate is headed toward Phase I clinical trials for infections due to extended-spectrum β-lactamase-producing Enterobacteriaceae.—RICK MULLIN


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter