Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Drug Discovery
Amgen unveils its KRas inhibitor in human clinical trials
AMG 510 shuts down a mutant version of the cancer target via covalent interaction
by Bethany Halford
April 3, 2019
| A version of this story appeared in
Volume 97, Issue 14

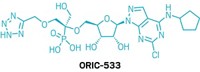
Drugmaker Amgen revealed the structure of AMG 510—the first covalent inhibitor of a mutant form of the cancer-target KRas to make it into human clinical trials. The molecule was disclosed at the American Chemical Society national meeting in Orlando on Wednesday during a session of the Division of Medicinal Chemistry.
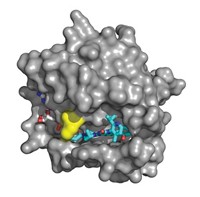
KRas proteins relay signals for cells to grow and divide or to mature and take on specialized functions. Mutants of the proteins are common in many cancers. Because of their critical functions, they’ve been eyed as cancer targets for years, but the spherical protein’s landscape is as featureless as billiard ball when in its active state, making it tough for drugmakers to find a place to slip in a compound that might muck up the protein’s inner workings. “There’s almost nowhere that a drug can stick to on that protein,” said Amgen’s Victor Cee, who presented the compound.
About 14% of non-small-cell lung cancer adenocarcinoma patients and roughly 5% of colorectal cancer patients possess the KRas mutant known as KRas G12C, so named because the glycine at its 12–position is mutated to a cysteine. That mutation provides a handhold for drugmakers, Cee explained, because the cysteine’s sulfur can make an irreversible, covalent bond to a drug, allowing the molecule to prevent the protein from doing its job. Making that covalent bond, he said, “gives us extraordinary specificity for just the mutant protein, which is important for things like tolerability.”
Cee and his colleagues tackled KRas G12C by looking for molecules that might latch on to its inactive form, which, unlike the active form, has some shallow binding pockets. “We felt that in the inactive state it kind of lets its guard down and there was a chance of getting something in there next to the cysteine that would react with it,” he said.
The researchers screened a series of 663 acrylamides and found one that weakly bound to KRas G12C. Crystallographic data revealed that it sat in a shallow pocket of the protein. In collaboration with Carmot Therapeutics, the Amgen team began screening more potential inhibitors and again found many molecules that bound within this pocket in different ways. For some of these, crystallographic data showed that a histidine residue could flip up to reveal a hidden groove.
As the team further explored mutant-binding compounds, they found that the best performers were also able to shimmy into this groove by flipping out that histidine. Their work eventually led them to AMG 510. This molecule’s methyl-, isopropyl-substituted pyridine ring gets stuck in one of two conformations, making AMG 510 an atropisomer. One of these atropisomers is more active, Cee said. It makes almost 25 van der Waals interactions with the KRas G12C mutant.
AMG 510 isn’t the only compound in human clinical trials that targets KRas G12C. Mirati Therapeutics began a trial with its covalent inhibitor MRTX849 early this year. But that molecule’s structure has not been disclosed yet.
A few days before Cee’s talk at the ACS meeting, Amgen presented preclinical data on AMG 510 at the American Association for Cancer Research (AACR) meeting. The company reported that AMG 510 can clear colon cancer from mice when given in combination with checkpoint inhibitors, which unleash the body’s immune system to attack cancer.
“Amgen presented very solid preclinical data for AMG 510, especially in combination with checkpoint inhibitors,” said Nir London, who works on drug discovery at the Weizmann Institute of Science and saw Amgen’s presentation at AACR. “It’s amazing to see how fast this story evolved,” he said, noting that the first proof-of-concept covalent KRas inhibitors came out of Kevan Shokat’s lab at the University of California, San Francisco just six years ago. Amgen’s work, London added, is “a shining example of the potential of rationally designed covalent inhibitors.”


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter