Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Drug Discovery
Open-source drug discovery takes aim at malaria and neglected diseases
Global collaborations share molecules and data to bring medicines to the developing world
by Mark Peplow, special to C&EN
January 29, 2019
| A version of this story appeared in
Volume 97, Issue 5

When an unremarkable stack of 96-well plates arrived in Fabrice F. Boyom’s lab in 2012, he couldn’t have known how the molecules stored inside would transform his research. Not only did the compounds accelerate his efforts to find treatments for tropical diseases, but they also attracted grants that furnished his lab with essential equipment and sparked international partnerships. “We have been able to develop collaborations across the world for drug discovery,” says Boyom, a biochemist at the University of Yaoundé I, in Cameroon. “The improvement has been huge.”
The plates were part of the Malaria Box, packaged and shipped by the Medicines for Malaria Venture (MMV), a nonprofit drug research foundation based in Geneva. Between 2011 and 2015, MMV distributed hundreds of these boxes, free of charge, to almost 200 research groups in 30 countries. The goal was to make it easier for academic researchers to discover new malaria treatments among the compounds stored within. The only condition was that they should put their results in the public domain.
And they did. Their work identified promising compounds that attacked the Plasmodium falciparum malaria parasite and that have led to further drug development work (PLOS Pathog. 2016, DOI: 10.1371/journal.ppat.1005763). But the campaign also found molecules that hit other disease agents. Boyom, for example, found a piperazine acetamide (MMV007791) that is active against the parasite that causes toxoplasmosis, a condition that can rob children of their sight, and he is now making derivatives of the compound to enhance its activity (Antimicrob. Agents Chemother. 2014, DOI: 10.1128/AAC.02541-14).

The project also revealed a huge appetite among academic researchers for libraries of promising compounds that they could search for treatments against neglected tropical diseases. So MMV, funded in part by the Bill and Melinda Gates Foundation, followed up in 2015 with the Pathogen Box, shipping out hundreds of compounds selected for their potential activity against the parasites that cause diseases like schistosomiasis and human African trypanosomiasis (sleeping sickness).
This week, MMV launches its latest box of coveted compounds. The Pandemic Response Box provides researchers with a selection of antiviral, antibacterial, and antifungal leads for screening against diseases, including those caused by the Zika and Ebola viruses. Boyom is particularly excited about this one: “Of course, I will request the box,” he says.
MMV’s boxes demonstrate the growing momentum behind more “open” approaches to drug discovery for malaria and neglected tropical diseases, in which researchers publicly share their data about therapeutic candidates. This openness is a radical departure from the traditional model of commercial drug research, in which intellectual property is zealously guarded, and advocates say that this change is sorely needed.
Many of the diseases endemic to developing countries take a huge toll on global health, yet they inspire relatively little research by the pharmaceutical industry. Developing a new drug is enormously expensive, and companies need to justify that investment to their shareholders. But the people affected by tropical diseases are usually far too poor to pay for pricey treatments, and global aid budgets struggle to cover the costs.
“The industry is producing some amazing drugs, but there are a lot of areas where drugs will never be forthcoming, given the economic constraints,” says Matthew H. Todd, chair of drug discovery at University College London. To put it bluntly: inventing drugs for poor people just isn’t profitable.
Open pharma projects are now making headway in tackling this problem, with some helping deliver compounds that are progressing through clinical trials. At the same time, they are sparking plenty of debate about how much openness is required, or even desirable, to truly be considered open projects. Some projects include any researchers who want to participate, share all their data online, and completely eschew patents. Others work more like conventional research collaborations—researchers cannot simply declare that they are joining in—and although all the preclinical work is published in open-access papers, the best drug candidates are typically patented before they proceed to clinical trials.
Despite the strategies’ differences, they all share an ambition to mobilize the global research community on behalf of patients whom conventional business models have failed. “The key to all of them is partnership,” says F. Javier Gamo, biology director for global health at GlaxoSmithKline’s research facility in Tres Cantos, Spain.
Boxes of opportunity
Malaria caused 435,000 deaths worldwide in 2017, 61% of which were children under the age of five, according to the World Health Organization. The best weapons against P. falciparum are called artemisinin combination therapies, but the parasite’s growing resistance to these drugs means that new therapies are urgently needed.
Roughly two-thirds of funding for research and development on neglected diseases comes from the public sector, predominantly from the governments of high-income countries, while the private sector contributes only 16%. Source: Policy Cures Research G-FINDER report 2018.

In an effort to stimulate research into new treatments, three groups collaborated with MMV to release a glut of data in 2010. After screening more than 2 million compounds, one of the groups, from GSK, published a list of 13,500 molecules with antimalarial activity, along with details about their potency, toxicology, and other properties (Nature 2010, DOI: 10.1038/nature09107). “We really changed the way we think about intellectual property and how we work with others,” Gamo says.
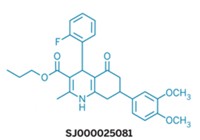
Around the same time, a consortium led by St. Jude Children’s Research Hospital released a similar catalog of malaria drug leads (Nature 2010, DOI: 10.1038/nature09099). And so did a third consortium, led by Novartis.
All told, these efforts provided a list of about 20,000 potential antimalarials that are logged in ChEMBL, an open chemical database curated by the European Bioinformatics Institute. That’s a great starting point for drug discovery, but many academic researchers wanted physical samples of the molecules for testing. “Most of the people who do this are not chemists and had no idea where to get the compounds from,” says Timothy Wells, chief scientific officer at MMV. These requests led MMV to collect 400 of the most promising compounds into the Malaria Box (PLOS One 2013, DOI: 10.1371/journal.pone.0062906).
MMV also realized that some of the researchers who requested these boxes did not have the resources to properly test them, so the organization provided some teams with grants to support their work. Boyom won $200,000 in funding from MMV, which he used to equip his lab with biosafety cabinets, a fluorescence microscope, and more. “That was really critical for us to be able to perform these studies,” Boyom says.
Unlike the compounds in MMV’s Malaria Box, those in the new Pandemic Response Box—a joint project with the Drugs for Neglected Diseases initiative (DNDi)—were selected by searching chemical databases for compounds previously reported as antibacterials, antifungals, and antivirals. Medicinal chemists from MMV, DNDi, and other organizations settled on a list of promising-looking compounds that represented a wide range of chemical families. MMV bought some of the compounds from chemical suppliers and had a contract research organization synthesize the rest. MMV is not prescriptive about how researchers test the compounds, as long as they share their protocols along with the results. If multiple researchers hit upon and test the same compound from the boxes, MMV sees this as confirmation of results rather than duplication of effort. And the organization tries to bring together groups working on the same hit, potentially forming partnerships. “It provides an opportunity to share best practice and build a research community,” Wells says.
Line of attack
Open research on MMV’s compound boxes has not yet yielded any molecules that have made it into clinical trials. But MMV is not the only organization mining the bonanza of antimalarial leads released in 2010. The companies involved are part of wider consortia that are also pursuing compounds identified by these screening efforts, and many are doing so in a more open manner than usual. GSK, for example, allows external researchers to work with company scientists at its Tres Cantos facility on malaria and neglected diseases.

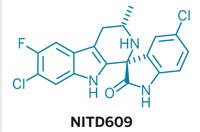
Using their 2010 list, scientists at Tres Cantos have developed a pyrrolidinamide antimalarial called GSK701. In August 2018, the company presented results from animal tests at the American Chemical Society national meeting in Boston that suggested that GSK701 was about as effective as artemisinin-based therapies, and the company is now preparing for a clinical trial. Meanwhile, work on the Novartis library has also borne fruit. In collaboration with MMV, for example, the company launched a Phase IIb clinical trial in 2017 of an imidazolopiperazine antimalarial called KAF156. Last year, Novartis also began a Phase IIa trial of cipargamin, otherwise known as KAE609, a spiroindolone antimalarial.


Researchers are also carrying out fresh screening campaigns to boost the pipeline of potential antimalarial leads. In December, a team including GSK, Novartis, MMV, and academic researchers unveiled a list of compounds that can hit Plasmodium parasites at a different stage of their complex life cycles than other drugs (Science 2018, DOI: 10.1126/science.aat9446). Most malaria therapies stop the parasites from replicating in a victim’s bloodstream, but only after a person is already experiencing malaria symptoms. The compounds identified in the new screen take aim at an earlier stage of the life cycle, when the immature parasite is just bedding down in the host’s liver. Small-molecule drugs derived from these compounds could prevent people from developing full-blown malaria rather than simply treating them once they fall ill. “If you could protect people from malaria with a patch or a long-lasting injectable, you could think about breaking the cycle of transmission,” says Elizabeth A. Winzeler at the University of California San Diego, who led the work.
Advertisement
The researchers screened about half a million compounds against immature parasites extracted from the salivary glands of hundreds of thousands of mosquitoes. “We used UCSD undergraduates—they’re happy to make $11 per hour by coming in on Wednesday mornings to dissect mosquitoes,” Winzeler chuckles. That herculean effort ultimately produced a short list of 631 promising antimalarials.
The effort is a “tour de force,” says Margaret A. Phillips, a biochemist at the University of Texas Southwestern Medical Center who was not involved in the work. All the hits and associated data have been published openly, so any research team can work to identify the proteins being hit by the compounds, understand the compounds’ mechanisms of action, and fine-tune them to enhance their activity. “It provides a way for the whole community to drive forward both the chemistry and the biology,” Phillips says.
Open wide
These efforts are certainly more open than traditional drug-discovery programs. Yet many philanthropic funders also encourage researchers to patent compounds once they approach clinical trials because the funders want reassurance that intellectual property infringements aren’t going to derail the costly process they’ve championed. “They absolutely want to know that if we develop a drug, we will be able to launch it,” MMV’s Wells says.
In contrast, some researchers advocate a greater degree of openness, modeled after open-source software development. Take Linux, an operating system that can be modified and distributed freely by anyone, for example. Its progress has relied on a community of developers working collaboratively on the underlying code, without any intellectual property restrictions.
The idea of applying those principles to drug discovery emerged around 2004 (PLOS Med. 2004, DOI: 10.1371/journal.pmed.0010056). University College London’s Todd argues that for a drug development project to be truly open source, it should have no barriers to participation and no patents. Crucially, participants also share their data and ideas publicly as they are working, just like software developers. Todd believes that this approach should ultimately offer a more efficient way to develop drugs than conventional “walled” research projects (PLOS Med. 2017, DOI: 10.1371/journal.pmed.1002276).
Although many projects are described as open source, Todd adds, relatively few actually adhere to all these principles. Wells acknowledges that Todd’s approach takes open-source drug discovery to a new level: “Matt is one step further down the open curve than most of our collaborators, and we’re interested in seeing how many people will follow him.”
Todd leads the Open Source Malaria project, which has already worked through two families of arylpyrrole compounds from GSK’s 2010 screen (ACS Cent. Sci. 2016, DOI: 10.1021/acscentsci.6b00086). The compounds were ultimately a bust, so the project has pivoted to a series of triazolopyrazines identified in a Pfizer compound library that was screened by MMV and partners. The project’s latest experiments in mice show that the best molecules are highly potent and have low toxicity, although their solubility needs to be improved. “The aim is to get one of these molecules into clinical trials,” Todd says.
Last year Todd helped launch the Open Source Mycetoma project, whose collaborators include DNDi. Fungal mycetoma is caused by the fungus Madurella mycetomatis, which creates sores and swelling that can be so severe they render victims’ limbs unusable. Existing drugs are expensive, toxic, and not very effective, and frequently the only available treatment option is amputation, to prevent the infection from spreading to other parts of the body. The project aims to develop new treatments through an open-source collaboration and already has some promising leads from a screen of MMV’s Pathogen Box (PLOS Neglected Trop. Dis. 2018, DOI: 10.1371/journal.pntd.0006437). Very little is known about the biochemical mechanism of the disease, so targeted drug design isn’t currently an option, says Wendy W. J. van de Sande of Erasmus University Medical Center, who works on the mycetoma project. “For mycetoma, screening libraries of compounds is the most straightforward approach,” she says, adding that she is looking forward to testing the compounds in the Pandemic Response Box for their antifungal activity.
No medicine has ever made it all the way through an open-source process like this, from discovery to patient, but Todd notes that open-source drug-discovery projects have already overcome some key stumbling blocks. Electronic laboratory notebooks and specialized web platforms have smoothed online collaboration, while academic journals are increasingly willing to publish papers whose data have already been shared in the public domain (as preprints, for example) and make them open access.
One challenge for open-source drug discovery that hasn’t been entirely overcome is money. “The funding landscape is the biggest barrier, not just for open-source research but for any research,” says Christine Årdal, a health policy researcher at the Norwegian Institute of Public Health. According to the latest G-FINDER report, produced by the Policy Cures Research think tank, about $3.6 billion was invested in neglected-disease R&D in 2017, with just 16% of the funds coming from the private sector.
Raising funds for preclinical research is hard enough, but clinical trials require much larger investments. For example, it cost more than $500 million to put the Mosquirix (or RTS,S) malaria vaccine through its clinical trials. That’s one reason why companies use patents to protect their intellectual property—they need to ward off competitors to ensure a decent return on the substantial investment they’ve made in the trials.
But open-source champions like Todd—who has received funding from the Australian government, MMV, and other charitable foundations—argue that patents promote secrecy, stifle collaboration, are costly to enforce, and ultimately raise the costs of medical therapies. “The dominance of patents has negative consequences for the industry and for the rest of us,” agrees the University of Michigan’s Rebecca S. Eisenberg, a legal scholar who specializes in intellectual property in pharmaceutical research.
Todd argues that an open-source model could reduce the costs of clinical trials but acknowledges that it would still require substantial investment. So how can an open-source project fund expensive clinical trials without protecting its intellectual property? Aled M. Edwards thinks he has the answer. He is the CEO of the Structural Genomics Consortium, which has determined the structures of more than 1,500 medically relevant proteins and put them in the public domain.
In 2017, Edwards helped launch M4K Pharma, which claims to be the world’s first open-science drug-discovery company. “By open science, I mean no patents,” Edwards says. M4K aims to develop the first-ever medicine to treat diffuse intrinsic pontine glioma, a rare and fatal brain cancer that affects children.
The company is putting all its research in the public domain to facilitate open-source science, and academic collaborators who give their time to the project will benefit through their publications and research partnerships. The company receives funding from government agencies, foundations, and corporate donors, and it is owned by a charity that will reinvest any profits into tackling diseases through open science. Edwards compares it to the business model used by Newman’s Own salad dressing, which gives all profits to a charitable foundation. “If they can do it for salad dressing, why can’t we do it for medicines?” he says.
M4K will rely on regulatory exclusivity, rather than patents, to ensure there is enough financial incentive for a manufacturer to make and distribute the drug. Some drug regulators grant companies several years of market exclusivity when they approve a drug—the US Food and Drug Administration typically offers five years for a new chemical entity, seven years if the drug treats a sufficiently rare disease, and an extra six months if it is a pediatric drug. That period of exclusivity should be enough to secure a payback, Edwards says. And whereas patents can be invalidated in court (and frequently are), “regulatory exclusivity is a slam dunk; you can’t overturn it,” he says. Edwards is confident that potential competitors are more likely to work with M4K than try to scoop the firm, adding that because the data will be in the public domain, rivals cannot patent the results anyway. If M4K wins approval for a drug, it could license the drug to a manufacturer on the condition that the drug be affordable.
In principle, the business model could work, Eisenberg says. But she doubts that it will become the norm for drug discovery in general. “It would be very difficult to get the pharmaceutical industry to give up its patents,” she says.
Todd hopes that discussions at the Global Open Source Pharma Conference, being held in Paris in March, can yield potential solutions to the economic challenges of bringing an open-source medicine through clinical trials. But for now, Årdal says, open pharma projects of any stripe are a welcome step, and efforts like MMV’s Pandemic Response Box provide an important way to boost participation. “We need to get more scientists involved,” she says, “because there’s simply not enough research going on for these neglected diseases.”
Mark Peplow is a freelance writer based in the UK.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter