Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Support nonprofit science journalism
C&EN has made this story and all of its coverage of the coronavirus epidemic freely available during the outbreak to keep the public informed. To support us:
Donate Join Subscribe
Vaccines
Covid-19
What will it take to make an effective vaccine for COVID-19?
Scientists are optimistic about our chance of success, but with a wide range of technologies and strategies in play, not all vaccines are alike
by Ryan Cross
July 17, 2020
| A version of this story appeared in
Volume 98, Issue 28

In brief
COVID-19 vaccines are being developed with a previously unimaginable urgency. More groups are working faster than ever before to develop shots that will protect us from the novel coronavirus, and hopefully bring an end to the pandemic. At first glance, the more than 160 vaccine programs seem remarkably similar, mostly focused on inducing immunity to the coronavirus spike protein. A closer look reveals many differences, including the types of vaccine technologies deployed, how the spike protein is modified and displayed to our immune systems, and the kinds of immune responses these different approaches will elicit.
There was a moment, just over 200 days ago, when we’d never heard of a coronavirus, when everything we did wasn’t shrouded with the specter of COVID-19. We crammed into living rooms, sang, danced, clinked glasses, showed 2019 out the door. We eagerly welcomed the new decade, filled calendars, and planned trips. Hugs and handshakes weren’t a health threat. Walking past someone in a crowded grocery store wasn’t anxiety inducing. Pictures of crowded beaches and bars didn’t evoke anger. How the world has changed.
In early January, no one could have known how truly catastrophic this novel coronavirus would be. Yet before this particular virus, SARS-CoV-2, was discovered, a few prescient people had already begun preparing for it. For decades, virologists have warned of an impending pandemic. We’re overdue, they said. Some groups even had the foresight to begin developing vaccines for a different coronavirus. Once SARS-CoV-2 emerged, those groups had a template to begin making vaccines for the yet-to-be-named disease, COVID-19.
As the pandemic grew, other companies and academic teams started working on their own vaccines for COVID-19. By early April, more than 100 programs were reportedly underway. Even then, vaccines remained a distant prospect. Amid shutdowns and social distancing, we simply yearned for summer, for a break from the virus. The reprieve never came.
Vaccines, for all intents and purposes, were the backup plan. Now, we need them more than ever.
“Without a vaccine, I don’t think we can put a lid on this,” says Paul Young, a virologist developing a COVID-19 vaccine at the University of Queensland. “It will continue to be a fire that rages through the world for quite some time until literally everyone is infected unless we are able to intervene.”
For nearly 7 long months, SARS-CoV-2 has pushed us to our limits. By mid-July, the virus had infected more than 13 million people and killed more than 580,000. About a quarter of those recorded cases and deaths belong to the US, a fraction likely to rise as so many Americans seemingly give up on the simple public health precautions that other countries have used to curtail the spread of the virus.
Yet there is cause for hope. In those same 7 short months, scientists have made strides that might normally take 7 years. Companies are beginning large trials with tens of thousands of people this month to see if their experimental vaccines can prevent disease. The pandemic has spurred the fastest vaccine development programs in history. While some groups are pushing to have vaccines available this winter, maybe sooner, others think such timelines are preposterous, and potentially reckless. Many questions remain, but there are two things that nearly everyone can agree on.
First, we need a vaccine to end this pandemic. “There is no doubt,” says Daria Hazuda, vice president of infectious diseases discovery at Merck Research Laboratories. “Given how widespread this is globally I just don’t think it is going to go away by itself.” Second, scientists are confident that at least one vaccine, and hopefully more, will eventually work. “There is every reason to believe that we can make a vaccine against this kind of virus,” says Paul Offit, a pediatrician and director of the Vaccine Education Center at Children’s Hospital of Philadelphia. “I think it is very likely that we will have an effective vaccine by the middle of next year,” he adds.
From that consensus, however, opinions diverge.
On the surface, all COVID-19 vaccine candidates have the same goal: generate an immune response that protects you from the virus. But under the hood, these vaccines use a range of technologies—from tried and true to new and untested—to teach our bodies how to defend itself against the virus.
This summer, C&EN interviewed more than three dozen scientists, doctors, and business leaders to illuminate the complementary, and occasionally conflicting, strategies employed by groups developing the most advanced and well-funded COVID-19 vaccines. There’s much to learn still, and more definitive answers will come in time, but we already know the questions we need to be asking to make an effective vaccine.
Here’s how we get back to normal.
I. How hard is it to make a vaccine against a virus?
Scientists have devised many ways to protect against an infection. In mid-July, the World Health Organization had counted 23 COVID-19 vaccine programs in clinical testing, and another 140 in preclinical development. “This is just an unprecedented effort, every possible vaccine strategy is being used, including ones that have never been used before,” Offit says.
The most traditional approach to making a vaccine is to simply use the virus itself, allowing your immune cells to learn how to fight it without you actually having to suffer through the disease. Viruses can either be left “alive” but attenuated—where scientists take all the chutzpah out of it—or they can be “killed” with chemicals and heat that leave them unable to replicate. Historically, some of the most effective vaccines, such as those for measles, polio, and smallpox are attenuated or inactivated vaccines.

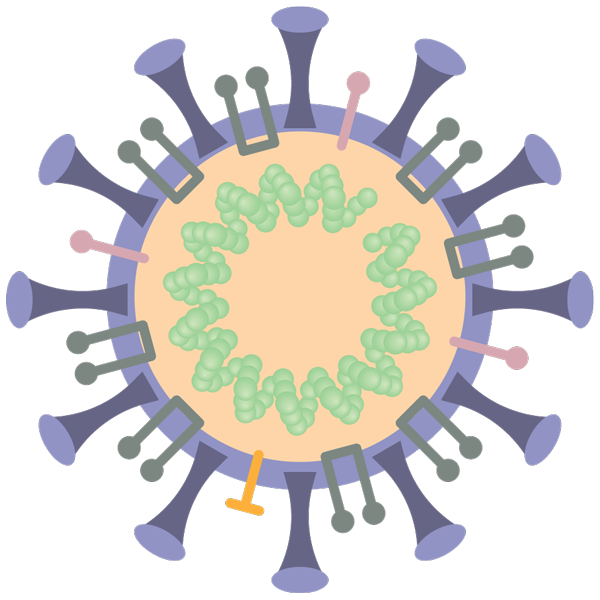
Today, the most popular approaches for making new vaccines all focus on isolating the specific part of the virus believed to be most important for immunity. For SARS-CoV-2, that part is incontrovertibly the spike protein—immediately recognizable in cartoons of the virus as the mushroom-like knobs studding its spherical surface. The coronavirus uses these spike proteins to grab hold of a human protein called ACE2, the first step in an infection. Nearly every COVID-19 vaccine candidate shares the objective of trying to prevent this interaction between the spike protein and ACE2.
Giving our immune cells target practice with a harmless form of the spike protein should allow them to halt the real virus in its tracks. A large number of groups are working on making the spike protein itself, detached from the virus, as the primary vaccine ingredient. Genetic engineering allows scientists to easily copy and paste the genetic code of the spike protein into cells that are optimized to grow in large vats, where they crank out large quantities of the protein. Vaccines for hepatitis B, shingles, and other diseases are made with this approach, which yields what’s known as a subunit protein vaccine.
But developing manufacturing processes for any of these more traditional vaccines typically takes months, if not much longer. Making attenuated or inactivated vaccines requires special facilities with extra safety precautions to grow large numbers of the actual virus, while subunit protein vaccines require scientists to optimize cells that can make the viral protein and then patiently wait for the cells to multiply.
Recently, there’s been growing excitement for experimental vaccines that take a different and faster route. Based on newer technologies, these vaccines simply contain the genetic code for the spike protein, and come in several forms, including DNA, messenger RNA, and viral vectors—where a harmless virus is rejiggered into a gene-delivery vessel. But the end goal for all of them is the same: transport the genetic instructions for the spike protein into human cells in order to temporarily turn those cells into spike protein factories. No DNA or mRNA vaccines have ever received regulatory approval, and only two viral vector vaccines—both to prevent Ebola virus—have been licensed for humans.
Frank DeRosa, the chief technology officer of the mRNA company Translate Bio, explains that mRNA vaccines let our own cells make the spike protein just like they would if we were infected with the real virus. These vaccines, along with DNA and viral vector vaccines, allow the spike protein to be trafficked to the cell membrane surface where it is displayed, or else chopped up and presented in pieces to immune cells. “You are just letting the body do what it would do normally,” DeRosa says. “That’s one of the advantages of mRNA.”
Gene-based vaccines should also allow the protein to undergo glycosylation, a cellular process of tacking sugars onto the protein in specific patterns, which will give the immune system a more accurate mug shot of the spike protein. These sugar patterns can differ in subunit proteins, depending on the kinds of cells used to manufacture them.
Genetic vaccines have another key advantage: they are breathtakingly fast to design and produce. The only thing that changes significantly between two genetic vaccines is the segment of code being delivered. “The manufacturing process for one RNA is a lot like the manufacturing process for another RNA,” says Phil Dormitzer, Pfizer’s chief scientific officer for viral vaccines. The same is largely true for DNA vaccines, and true to a lesser degree for viral vector vaccines. It’s why most of the fastest moving programs for COVID-19 are gene-based vaccines.
The current record speed for making a modern vaccine is Merck’s viral vector vaccine for Ebola, which took 5 years to design, test, and earn government approval. For COVID-19, many companies say that process could be collapsed into a year or two. Some firms, including AstraZeneca, Moderna, and Pfizer, expect to have efficacy data this fall, and the US government plans to preorder 300 million doses ready for distribution by January 2021.
Those timelines have plenty of skeptics. “The notion that we can have something done by the fall is frankly ludicrous, that is just not going to happen,” says Kenneth Kaitin, director of the Tufts Center for the Study of Drug Development. “I would suspect that by this time next year we are still going to be looking forward to when that first vaccine hits the market.”
Vaccine variety
More than 160 vaccines are in the works to prevent COVID-19. Here are the major types of technologies being used to make them.
-
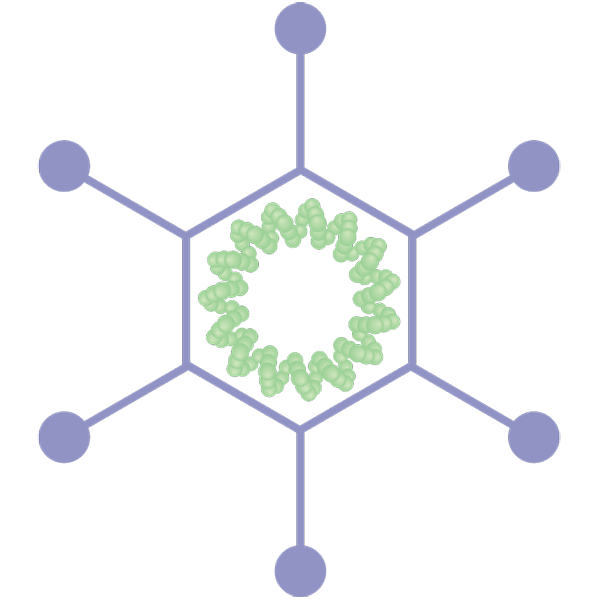
Attenuated and inactivated virus vaccines
Attenuated virus vaccines contain a “living” but weakened version of SARS-CoV-2. Inactivated virus vaccines contain SARS-CoV-2 that has been “killed” with heat or chemicals like β-propiolactone or formalin. Several childhood vaccines are attenuated or inactivated virus vaccines.
-
Subunit protein vaccines
Subunit protein vaccines contain the SARS-CoV-2 spike protein, which the virus uses to enter human cells. These vaccines often include adjuvants, which are molecules that stimulate the innate immune system to help simulate a natural infection. More groups are developing subunit protein vaccines for COVID-19 than any other technology.
-
Viral vector vaccines
Viral vector vaccines use a different virus—such as the adenovirus, measles virus, or vesicular stomatitis virus—that is genetically engineered to carry the gene for the SARS-CoV-2 spike protein, which will be made by our cells. Viral vector vaccines for preventing Ebola have recently been approved, but others are still experimental.
-

Nucleic acid vaccines
Nucleic acid vaccines encode genetic instructions for the SARS-CoV-2 spike protein into DNA, delivered into our cells with an electric shock, or RNA, delivered into our cells via a lipid nanoparticle. These vaccines can be rapidly designed and manufactured, but no DNA or RNA vaccine has been approved for humans.
II. Does the immune system view all vaccines equally?
Most vaccine developers believe that the potential protection offered by these vaccines hinges on teaching our immune cells to make the right kind of antibodies. In theory, antibodies can bind to any part of the spike protein, but only certain ones, the so-called neutralizing antibodies, bind to the spike protein in a manner that prevents the virus from infecting our cells.
“Neutralizing antibodies are the most important biomarker to follow in the vaccine studies, and higher the antibody titers, the better,” says John Shiver, senior vice president for global vaccines R&D at Sanofi.
You might imagine that the best way to get those high levels of neutralizing antibodies is to simply present the spike protein in its most natural form. But the spike protein is a wily shapeshifter, and many groups think that tweaking the spike protein will be necessary to induce a good neutralizing antibody response.
After the spike protein binds to ACE2, it undergoes a dramatic transformation. A spring-loaded portion of the spike shoots into the human cell membrane and then pulls the virus and cell so close together that their membranes fuse. This allows the virus to spill its genes and guts into the cell, where it begins replicating.
So scientists think there are probably two major ways an antibody can prevent infection: it can either directly block the spike’s interaction with ACE2 in the first place, or it can gum up the spike’s spring-loaded machinery and impede its fusion with our cells.
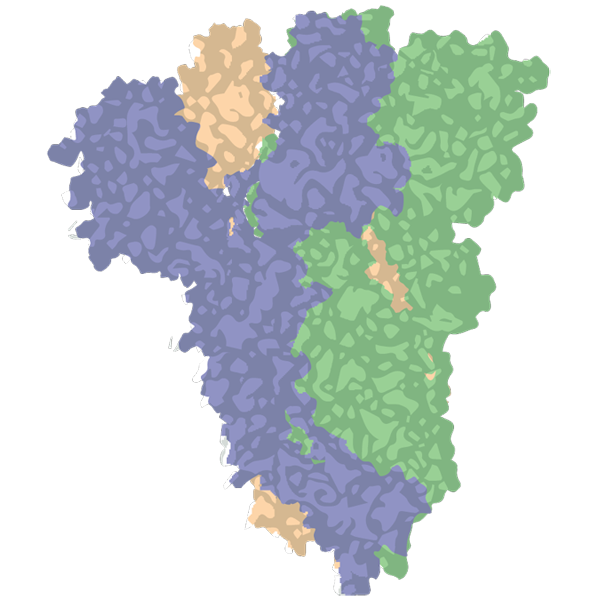
In 2016, while scientists were studying the spike protein of a different coronavirus, they discovered that embedding two prolines—the most rigid of amino acids—in a particular part of the spike helped lock it into the shape that it takes before binding ACE2. Many researchers believe it is crucial to show your immune cells this so-called prefusion form of the spike protein in order to make antibodies that prevent infection. In contrast, if the vaccine teaches the immune system to make antibodies to the postfusion form, the shape the spike protein takes after binding to a cell, those antibodies will bind to the spike too late to prevent infection, says Andrew Ward, a structural biologist at Scripps Research who co-led the study.
Before the pandemic, that double proline mutation, called the 2P mutation, proved generalizable to several coronavirus spike proteins. So when SARS-CoV-2 emerged in early January, researchers were able to quickly add this mutation into the design of a COVID-19 vaccine. The mRNA company Moderna and researchers at the National Institute of Allergy and Infectious Diseases (NIAID) made “a somewhat risky decision” to begin manufacturing a COVID-19 vaccine based on the virus’s spike sequence and the addition of the 2P mutation without any further experiments, explains Barney Graham, deputy director of the Vaccine Research Center at NIAID.
Since then the 2P mutation has made its way into subunit protein vaccines, mRNA vaccines, and viral vector vaccines. Jason McLellan, the scientist who discovered the 2P mutation, is now looking for other promising ones. His lab at the University of Texas at Austin has tested more than 100 additional mutations, which led to the creation of a novel prefusion spike protein dubbed HexaPro. It’s more stable, and, when plugged into an mRNA vaccine, causes cells to make 10 times the amount of spike protein. He says companies making COVID-19 vaccines are already testing HexaPro in lab studies, and his lab is working on further improvement. “We are always tweaking,” he says. “You can kind of do this forever but at some point you just have to pick something and move it forward.”

Other groups are making their own unique modifications to the prefusion spike. Scientists at the University of Queensland have made a subunit protein vaccine where the trimer of the spike is held together by what Queensland virologist Keith Chappell calls a molecular clamp. “It is gripped at the base, and the top has natural flexibility,” he says.
Other groups are forgoing the prefusion conformation in favor of a more natural, functional spike protein. That includes the DNA vaccine company Inovio Pharmaceuticals, which used this approach to elicit neutralizing antibodies in people who got its experimental MERS vaccine.
One of Merck’s two viral vector vaccines is based on vesicular stomatitis virus (VSV), also used to make the firm’s recently licensed Ebola vaccine, Ervebo. Unlike the adenoviral vector vaccines under development for COVID-19, which just carry the genetic instructions for the spike protein, the VSV viral vector is designed to display the SARS-CoV-2 spike protein on its surface, where it can be used to enter human cells. “It is kind of an authentic presentation,” says Christopher Parks, whose lab led the design of the vaccine at IAVI, before Merck said it would test it in humans.
Another strategy is to use just a key fragment of the spike protein. It turns out that the most potent neutralizing antibodies made by people who recover from COVID-19 almost always target a particular part of the spike protein. That key section, called the receptor-binding domain (RBD), sits at the top of the spike, where it makes direct contact with ACE2 on human cells. For this reason, some groups are developing vaccines that simply use the RBD—either made as a subunit protein or encoded in mRNA.
RBD-based vaccines could have the advantage of helping the immune system focus on developing neutralizing antibodies to the part of the protein that matters the most. It’s also a relatively small part of the large spike protein, which could make these vaccines cheaper to manufacture.
But its small size has drawbacks too. Scientists say we typically develop better immune responses against larger proteins. And researchers are starting to discover neutralizing antibodies that bind to other regions of the spike protein outside the RBD as well, ones that might work by halting the virus’s fusion to the human cell, rather than by blocking its binding to ACE2. In general, having neutralizing antibodies to multiple sites should limit the virus’s ability to mutate and escape neutralization.
One study in monkeys testing six different DNA vaccines all encoding various versions of the spike protein found that the full-length spike protein induced higher levels of neutralizing antibodies than the RBD. A small study testing four variations of subunit proteins in rabbits found the opposite: the RBD vaccine induced the highest levels of neutralizing antibodies.
“The RBD might be good enough. And when you are making a vaccine, you just need to make it good enough,” NIAID’s Graham says. But, he adds, “we just think it is not quite as good as the whole thing.”
Pfizer, which is working with the German mRNA company BioNTech, may be the only group that is hedging its bets by testing multiple vaccines in humans: two encoding the full prefusion spike protein and two encoding the RBD. “Although you can do plenty of testing preclinically, some questions you really have to answer in clinical trials,” says Pfizer’s Dormitzer.
If a particular paradigm proves most promising, it will be easy to construct a narrative about why one brilliant group had the right idea all along. You can reason your way into believing that any one front-runner vaccine will rise above the others just as easily as you can convince yourself that one approach is destined for failure. But as it stands, we don’t know which vaccines will work the best. Although animal studies can give clues about what won’t work in humans, the only way to determine how a vaccine will protect against infection is to test it in people.
III. How will our immune system protect us from the virus?
The need for speed
Key milestones in the rapid design, clinical testing, and funding of vaccines for COVID-19
January
January 10
- The first genome sequence of the novel coronavirus, later named SARS-CoV-2, is posted online.
January 13
- Moderna announces plans to develop an mRNA vaccine for the novel coronavirus.
January 23
- The Coalition for Epidemic Preparedness Innovations (CEPI) announces vaccine funding for Inovio Pharmaceuticals, Moderna, and the University of Queensland.
March
March 16
- CanSino Biologics and Moderna dose first volunteers in Phase I clinical trials of their vaccines.
March 17
- Pfizer announces partnership with BioNTech to develop and test multiple mRNA vaccines.
March 30
- Biomedical Advanced Research and Development Authority (BARDA) and Johnson & Johnson announce they are committing more than $1 billion to develop an adenoviral vector vaccine for COVID-19.
April
April 16
- BARDA awards Moderna up to $483 million to develop and manufacture its mRNA vaccine.
April 30
- AstraZeneca announces it will develop the University of Oxford’s adenoviral vector vaccine for COVID-19.
May
May 11
- CEPI commits $384 million to Novavax’s COVID-19 vaccine, its largest investment ever.
May 15
- US President Donald J. Trump announces Operation Warp Speed to supply 300 million vaccines to the US by January 2021.
May 18
- Moderna announces preliminary Phase I data from its vaccine trial via press release.
May 21
- BARDA says it will provide up to $1.2 billion for 300 million doses of AstraZeneca’s vaccine with the first shots arriving in October.
May 22
- CanSino publishes the first peer-reviewed data of a Phase I COVID-19 vaccine trial.
May 26
- Merck & Co. says it will develop two COVID-19 vaccines originally designed at Themis Bioscience—an Austrian company that it acquired—and IAVI.
May 29
- Moderna doses the first volunteers in its Phase II clinical trial of its mRNA vaccine.
June
June 20
- A Phase III trial testing the University of Oxford’s adenoviral vector vaccine begins in Brazil.
June 24
- The state-owned China National Pharmaceutical Group (Sinopharm) announces plans for a Phase III trial of its inactivated virus vaccine for COVID-19.
June 28
- 10 million people have been infected and 500,000 people have died from COVID-19.
July
July 7
- BARDA and the US Department of Defense sign a $1.6 billion contract with Novavax for 100 million doses of its vaccine.
The need for speed
Key milestones in the rapid design, clinical testing, and funding of vaccines for COVID-19
Jan. 10: The first genome sequence of the novel coronavirus, later named SARS-CoV-2, is posted online.
Jan. 13: Moderna announces plans to develop an mRNA vaccine for the novel coronavirus.
Jan. 23: The Coalition for Epidemic Preparedness Innovations (CEPI) announces vaccine funding for Inovio Pharmaceuticals, Moderna, and the University of Queensland.
March 16: CanSino Biologics and Moderna dose first volunteers in Phase I clinical trials of their vaccines.
March 17: Pfizer announces partnership with BioNTech to develop and test multiple mRNA vaccines.
March 30: Biomedical Advanced Research and Development Authority (BARDA) and Johnson & Johnson announce they are committing more than $1 billion to develop an adenoviral vector vaccine for COVID-19.
April 16: BARDA awards Moderna up to $483 million to develop and manufacture its mRNA vaccine.
April 30: AstraZeneca announces it will develop the University of Oxford’s adenoviral vector vaccine for COVID-19.
May 11: CEPI commits $384 million to Novavax’s COVID-19 vaccine, its largest investment ever.
May 15: US President Donald J. Trump announces Operation Warp Speed to supply 300 million vaccines to the US by January 2021.
May 18: Moderna announces preliminary Phase I data from its vaccine trial via press release.
May 21: BARDA says it will provide up to $1.2 billion for 300 million doses of AstraZeneca’s vaccine with the first shots arriving in October.
May 22: CanSino publishes the first peer-reviewed data of a Phase I COVID-19 vaccine trial.
May 26: Merck & Co. says it will develop two COVID-19 vaccines originally designed at Themis Bioscience—an Austrian company that it acquired—and IAVI.
May 29: Moderna doses the first volunteers in its Phase II clinical trial of its mRNA vaccine.
June 20: A Phase III trial testing the University of Oxford’s adenoviral vector vaccine begins in Brazil.
June 24: The state-owned China National Pharmaceutical Group (Sinopharm) announces plans for a Phase III trial of its inactivated virus vaccine for COVID-19.
June 28: 10 million people have been infected and 500,000 people have died from COVID-19.
July 7: BARDA and the US Department of Defense sign a $1.6 billion contract with Novavax for 100 billion doses of its vaccine.
All these vaccine efforts are grounded in the notion that producing high levels of potent neutralizing antibodies will prevent the virus from infecting our cells. Measuring those antibodies, however, is fraught with challenges, and we don’t even know what levels we should aim for.
Methods used to quantify that neutralizing antibody response are imperfect. Researchers infect cells in a petri dish with either a real or artificial version of SARS-CoV-2 to see how much of the virus is blocked with a particular concentration of antibody-containing plasma. The real and artificial methods yield different results. And, although those results have been cited as rationale for moving COVID-19 vaccines into large, late-stage trials, there is no standard for how these measurements should be reported.
For instance, some groups report the level of neutralizing antibody that inhibits 50% of the virus, while others use higher bars of 80, 90, or 100%. “If you make antibodies that neutralize 90% of the virus, that may not be good enough,” NIAID’s Graham says. “You want a neutralization that is 100% effective.”
“The number you get depends on the specifics of the assay you run, so comparing one company’s numbers to another company’s numbers is tricky,” Pfizer’s Dormitzer says. “Until we really establish what a protective level of antibodies is, the numbers may be a relative yardstick, but they don’t tell you if you are going to have protection or not.”
So far, companies have been using as their baseline the levels of neutralizing antibodies found in convalescent plasma of people who have recovered from COVID-19. But research shows that COVID-19 survivors make relatively low levels of antibodies, and one small study suggests they might only stick around for 2 to 3 months.

Such studies suggest that a vaccine that mimics a natural infection is a pretty low bar. “Immunity equivalent to natural infection may not be enough for this virus. It might need to be higher,” says David Corry, an immunologist and allergist at Baylor College of Medicine.
On average, each coronavirus has a couple hundred spike proteins that it can use to grab onto a cell, so the number of neutralizing antibodies circulating in our bodies likely needs to be much higher than the number of viruses attempting to establish an infection. If the antibody levels are not high enough, we may end up with only partial protection—where we still get an infection, and might even be able to spread the virus to others, but would be safe from progressing to the most severe forms of COVID-19 that hospitalize people.
But even determining the level of antibodies needed to lessen the brutality of the disease is not straightforward. “A level of antibodies in one person might send them off without any symptoms at all, while the same level of antibodies in another person may still leave them very sick,” Scripps immunologist Dennis Burton says.
Advertisement
Some scientists say that partial protection is a fine goal for the first generation of COVID-19 vaccines. “If you can keep people out of the hospital, to me that is a tremendous success,” says Gregory Glenn, president of R&D at Novavax. Such vaccines could save lives, and in a hypothetical world where everyone is vaccinated, most individuals could deal with mild cases of COVID-19, and society could return to normal.
Although vaccine makers have focused on neutralizing antibodies, this type of immune response might not last forever. In a study of 191 people tested for cold-causing coronaviruses over a period of 19 months in New York City, researchers found that 9 people were infected with the same virus twice, and 3 were infected with the same virus three separate times. We don’t know if either natural immunity or vaccines can prevent these kind of reinfections with SARS-CoV-2. One experiment showed that monkeys who were infected with high levels of SARS-CoV-2 were protected from reinfection 5 weeks later—although that study comes with the major caveat that monkeys don’t develop full-blown COVID-19 in the first place.
There’s reason to believe that other parts of the immune system, such as T cells, may be important for longer-lasting immunity. Scientists found that people who were infected with SARS-CoV-1, the virus that caused the eponymous severe acute respiratory syndrome (SARS) outbreak in 2003, still had neutralizing antibodies to the virus 2 years after infection, but not 5 years later. In contrast, researchers recently discovered that some people infected with SARS-CoV-1 back in 2003 still have T cells that recognize the virus all these years later.
While antibodies prevent viruses from infecting cells in the first place, T cells can spot cells that are already infected and selectively kill them, thereby halting the spread of the virus. T cells are also better than antibodies at targeting different parts of the virus. Antibodies target proteins on the outside of the virus, which for SARS-CoV-2 is the spike protein. Yet the spike is just one of 27 proteins encoded in the SARS-CoV-2 genome. The other proteins are located inside the virus, or are made by our own cells when the virus is replicating. T cells, unlike antibodies, can learn to spot molecular fingerprints of these proteins in virus-infected cells.
DNA vaccines and viral vectors are better at inducing T cell responses, while subunit protein vaccines primarily induce antibodies. The traditional attenuated virus vaccines that use a live virus—and therefore have all those internal proteins—are good at inducing both T cells and antibodies. “Every formulation or platform is different,” says Surender Khurana, a vaccine scientist at the US Food and Drug Administration. “These different platforms can have different kinds of immune responses, and we don’t know which immune response is most relevant.”
IV. How good is good enough for a COVID-19 vaccine?
Some vaccines, like the one for measles, provide lifelong immunity to nearly every single person who receives them. Others, such as flu vaccines, are needed every year, and even then sometimes only work 30% of the time. For COVID-19 vaccines, the FDA is aiming for something in-between those extremes. The FDA’s recently issued guidelines for COVID-19 vaccine development state that the agency expects a vaccine to either prevent disease, or reduce its severity, in at least 50% of vaccinated people.
Most vaccine developers are hoping for vaccines that work in a higher number, 70–90% of people. Although, as the FDA guidance suggests, there is more than one definition of what it means for a vaccine to work. “The home run is a vaccine that prevents severe illness, prevents mild illness, prevents spread, and that lasts a long time,” Pfizer’s Dormitzer says. Many people expect that the first wave of COVID-19 vaccines won’t hit all those marks. A vaccine might only prevent symptoms, but still allow the virus to replicate and spread. It may also just prevent severe COVID-19, but not mild illness or transmission.
If vaccines are not highly effective, their impact will be even further dampened if most people don’t take them. Surveys have suggested that 20–50% of Americans would not plan on getting vaccinated if a COVID-19 shot was approved. The polls raise serious questions about deploying a marginally effective vaccine. “It could end up making everything worse,” says Margaret Ann Liu, chair of the board and former president of the International Society for Vaccines. When the first antiretrovirals for HIV became available, many people lost their fear of the virus, and risky behaviors increased, she says. Something similar could happen for COVID-19. “So this is a very complicated question. It is not simply a question of efficacy.”
Another question is safety. Many of the most advanced vaccines are based on technologies that have never been used in large numbers of people. Before the pandemic, Moderna had tested its mRNA vaccines in only about 1,400 people. Now, it is gearing up for a 30,000 person placebo-controlled study in July. Likewise, the University of Oxford’s adenoviral vector vaccine—made from a virus that normally infects chimpanzees—had only been tested in 330 people across a dozen Phase I trials. By this fall, more than 40,000 people may have participated in a placebo-controlled clinical trial for Oxford’s COVID-19 vaccine based on the vector.
“We can make effective vaccines quite quickly,” says David Dowling, a vaccine researcher at Boston Children’s Hospital. “But safety is not something that can be measured in a single time point. It has to be observed over a period of time.” And a vaccine that is safe for one age group could be risky for another, he adds. Phase III studies should pick up uncommon side effects, but rare reactions likely won’t be known until the vaccine is approved and widely used. “Honestly, we may not have answers until years after a vaccine is deployed.”
Researchers warn that moving too fast could backfire. If one COVID-19 vaccine proves unsafe, it could drain public confidence in other vaccines for the disease. “There are potential risks with moving as rapidly as we want to move,” says Wayne C. Koff, CEO of the Human Vaccines Project. “The process of vaccine development normally takes a decade or more, and is being compressed into an 18-month window while we are still learning about the virus.”
Even if one or more vaccines are safe and work, they won’t necessarily protect everyone. Viral vector vaccines, for example, can be ineffective if given to someone previously infected with the same virus used to make the vaccine. And it’s an open secret that vaccines tend to work the worst for the people who need them most—the very young and the very old. “Traditionally vaccines were built for one-size-fits-all,” says Ofer Levy, director of the Precision Vaccines Program at Boston Children’s Hospital. His group, and Dowling, are studying how to make age-specific vaccines for COVID-19.
Vaccine developers are also quick to point out that no single company can do this alone. “We need several vaccines to work to end this pandemic,” says Sanofi’s Shiver, which is developing two COVID-19 vaccines based on different technologies. Sanofi, one of the largest vaccine makers in the world, expects to produce 1.3 billion doses of its COVID-19 vaccines in 2021, just a fraction of the 8 to 16 billion doses that may be needed to vaccinate the world, since many vaccines may require two shots. “I hope both of ours will work, and I hope our competitors’ vaccines will work, because there are a lot of doses to deliver to the world, and there is so much unknown,” Shiver says.
Advertisement
It is hard to overstate how much is riding on an effective vaccine. “A vaccine is the viable exit strategy that doesn’t involve millions of deaths,” says Peter Openshaw, an immunologist at Imperial College London. And even if an effective vaccine arrives, we must be prepared for the possibility that SARS-CoV-2 cannot be eradicated. “It may be possible to eliminate this virus with a global vaccination campaign, but it is too early to tell,” he adds. “There haven’t been many infections eliminated from the world.” The virus may be with us forever now.
Three previously unknown coronaviruses have jumped from wild animals into humans in the past two decades, all with deadly consequences. That’s prompted some scientists to start thinking ahead to the seemingly inevitable emergence of yet another new coronavirus. Ted Ross, director of the Center for Vaccines and Immunology at the University of Georgia, is working on developing a vaccine that is an amalgamation of the most immunogenic parts of several coronavirus spike proteins, including SARS-CoV-1, SARS-CoV-2, and coronaviruses that have yet to infect humans. “It contains the best of everything in the wild,” he says. He hopes to test the universal coronavirus vaccine in the clinic sometime in 2021.
“This virus is not going away. People still need to follow universal precautions like social distancing and wearing masks. We have to be careful,” Ross says. “We hope we will have a vaccine that works, and it is likely to happen, but we probably shouldn’t put everything in that basket. Therapeutics, antiviral drugs, and public health still need to be high on our priority list. We can’t wait for vaccines to come save us all.”
CORRECTION
On July 30, 2020, this story was updated to correct the number of doses Novavax is contracted to make. It is 100 million doses, not 100 billion.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter