Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Vaccines
Covid-19
The tiny tweak behind COVID-19 vaccines
Prepandemic coronavirus research by Jason McLellan and Barney Graham led to a trick for stabilizing the prefusion form of spike proteins
by Ryan Cross
September 29, 2020
| A version of this story appeared in
Volume 98, Issue 38

On Jan. 10, Chinese scientists uploaded the genetic sequence of a novel coronavirus, later named SARS-CoV-2, to an open-access website, GenBank. The virus had been linked to a growing number of mysterious pneumonia cases, and its rapid spread was beginning to raise alarms. A few hours later, Barney Graham woke up and saw the sequence. Though it was a Saturday, he got right to work.
Days before, his lab at the US National Institute of Allergy and Infectious Diseases (NIAID) had partnered with the biotech company Moderna to design an experimental vaccine for the virus, which causes the disease COVID-19. All they’d needed to start was that sequence.
Support nonprofit science journalism
C&EN has made this story and all of its coverage of the coronavirus epidemic freely available during the outbreak to keep the public informed. To support us:
Donate Join Subscribe
Six months later, Moderna began testing that COVID-19 vaccine in a Phase 3 clinical trial of 30,000 people. Results are expected later this fall.
Neither Graham nor Moderna could have known then how truly catastrophic the novel coronavirus would become. Two previous coronavirus outbreaks, which caused severe acute respiratory syndrome (SARS) and Middle East respiratory syndrome (MERS)—both deadlier but less contagious—were contained without global shutdowns and before vaccines could be developed. So how did they make the COVID-19 vaccine candidate so fast?
It helps that Moderna’s vaccine is based on messenger RNA (mRNA), a genetic molecule that scientists can synthesize more rapidly than the viral proteins or whole viruses used in conventional vaccines. Moderna and Graham’s lab also had a template to work from because they’d spent several years designing an experimental mRNA vaccine for the MERS coronavirus.
But there’s a third, more subtle secret to their success: a tiny but oh-so-important tweak to a critical viral component called the spike protein.
Viruses multiply by dumping their genes into our cells and hijacking our cellular machinery to crank out new virus particles. But first, they need a doorway into our cells. Coronaviruses are studded with spikes, which grab hold of proteins decorating our own cells like doorknobs. Once attached, the spike undergoes a dramatic transformation, stretching before partially turning inside out to forcefully fuse with our cells.
Scientists believe that for COVID-19 vaccines to be effective, our immune systems must develop antibodies that prevent this fusion. Such antibodies must target the spike protein in its aptly named prefusion conformation. Unfortunately for vaccine developers, spike proteins are liable to spring from their stubby prefusion shape into their elongated postfusion form on a hair trigger.
Fortuitously, Graham and a former postdoc, Jason McLellan, devised a solution to this problem before the pandemic. Through a bit of structural biology and persistent protein engineering, McLellan discovered that adding two prolines—the most rigid of the 20 amino acids—to a key joint of a vaccine’s spike protein could stabilize the structure’s prefusion shape. This 2P mutation worked in preclinical studies of Graham and Moderna’s MERS vaccine, so they applied it to Moderna’s COVID-19 vaccine.
As Norbert Pardi, an mRNA vaccine scientist at the University of Pennsylvania, puts it, we’re “very lucky, actually,” that scientists worked out the 2P mutation for a MERS vaccine before the COVID-19 pandemic. “It wouldn’t be possible to go so fast with the Moderna vaccine otherwise.”
Other companies, including Johnson & Johnson, Novavax, and Pfizer, are hoping the 2P mutation works for their COVID-19 vaccines too.
The 2P mutation might quite literally be the smallest detail that could make or break the first generation of COVID-19 vaccines. It’s an easy enough tweak to add during the early stages of vaccine design. And if successful, 2P-based vaccines may herald a new generation of vaccines whose molecular makeup is fine-tuned to craft a safer, stronger immune response.

Catching a shape-shifter
Vaccines are built on the premise that if you show immune cells a pathogen that they might one day encounter in the wild, they should learn to protect against the disease. Older vaccines present the immune system with an entire virus in a weakened or deadened form. More contemporary vaccines are made up of only a piece of the virus—a certain critical protein that should prompt the creation of antibodies that can shut down an infection. And the newest vaccines, like Moderna’s, deliver mRNA that encodes instructions for making that viral protein.
Yet sometimes showing the immune system the viral protein, in any of these forms, isn’t enough. Respiratory syncytial virus (RSV) is a prime example.

In the US, RSV sends an estimated 57,000 children under the age of 5 to the hospital each year; some 14,000 adults older than 65 die from the virus. There’s still no vaccine available.
In 1966, a decade after RSV was discovered, US National Institutes of Health researchers began testing an RSV vaccine made of a virus killed with formalin—an aqueous solution of formaldehyde. The trial was a disaster, McLellan says. Although infants who got the vaccine developed antibodies against the virus, they were not protected from infection. Instead, the vaccine seemed to make the disease worse. Some 80% of infants who got the shot were hospitalized after an RSV infection, compared with 5% of infants in the control group. Two vaccinated babies died from the infection. The tragedy tainted the RSV vaccine field for decades.
Scientists now know that RSV infects and fuses with human cells using its F protein, which, like coronavirus spike proteins, shape-shifts during infection. The F protein is far more unstable than spike proteins, and in 2016 Graham reported that formalin inactivation leaves the viruses coated with postfusion F. Our immune systems can make antibodies against postfusion F, but they aren’t very good ones.
The best antibodies prevent infection. These neutralizing antibodies are the goal of vaccine developers. Animal studies suggest that the initial RSV vaccines induced antibodies that bound to postfusion F but failed to neutralize the virus, leading to inflammation, clogged airways, and more severe disease than with no vaccine at all.
In theory, a vaccine based on the prefusion F should solve this problem.
RSV wasn’t the only virus that seemed impervious to vaccination. In particular, the inability to stop HIV with vaccines pushed biologists to start tinkering with the structures of viral proteins in an attempt to engineer a better immune response.
McLellan joined NIAID as a postdoc in Peter Kwong’s lab in 2008 to do that work with HIV, but he soon realized he was up against the hardest test case for these protein-engineering principles. Graham, who, as the deputy director of NIAID’s Vaccine Research Center, worked in the same building, encouraged McLellan to work with him on RSV. If they could find a way to modify the F protein and keep it locked in its prefusion form, they might have a shot at creating a successful RSV vaccine.

Figuring out exactly what the prefusion F protein looked like was their first challenge. McLellan and Graham decided to look for antibodies that neutralize RSV but don’t bind postfusion F. Such antibodies were likely binding to prefusion F, they reasoned, and could thus be used to lock the protein down. Doing so would allow McLellan, a trained X-ray crystallographer, to capture a snapshot of the prefusion F and antibody bound together. The team found three highly potent neutralizing antibodies that fit the bill.
The resulting picture explained why the older RSV vaccines had failed. The antibodies clung to the very tip of the prefusion F, a foothold that disappeared during the protein’s metamorphosis as it fused to its host cell.
The team embarked on a large protein-engineering effort to create more than 100 variants of F to find modifications that stabilized the prefusion form. McLellan found that swapping out two serines for cysteines introduced a disulfide bond that, like tape, helped hold the protein in place. The researchers made it even stabler with a second tweak—replacing two amino acids in its inner cavity with hydrophobic residues. “We created a vaccine molecule that couldn’t have been created without structural information,” McLellan says.
That molecule, called DS-Cav1 and published in Science in 2013 (DOI: 10.1126/science.1243283), induced 10 times the level of neutralizing antibodies as a vaccine with postfusion F did in a study with rhesus macaques. Several companies, including GlaxoSmithKline, J&J, Moderna, and Pfizer, are developing RSV vaccines that rely on prefusion-stabilization mutations.
“The RSV work showed that the protein sequence is not nearly as important as the protein conformation,” Graham says. In other words, a vaccine might contain or encode the right protein, but if that protein isn’t in the right shape, it won’t work. “This really changes the way you think about designing a vaccine.”
Stabilizing the spike
After early signs of success with the RSV vaccine, Graham and McLellan were eager to apply their techniques to another virus. As McLellan was starting his own lab at Dartmouth College in 2013, the recently emerged MERS coronavirus was top of mind. No one had ever solved the structure of a full coronavirus spike protein. These proteins are similar to the F protein on RSV but a whopping 2.5 times as large.
Despite many attempts, and even with outside help, the scientists couldn’t get the MERS spike protein to cooperate. So McLellan, Graham, and their collaborator at Scripps Research, Andrew Ward, turned to a different coronavirus, HKU1. The virus, which causes the common cold, was safer to work with and better at sitting still while its picture was being taken. In 2016, Ward’s lab used a technique called cryogenic electron microscopy to capture the structure of the HKU1 spike.
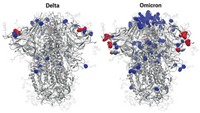
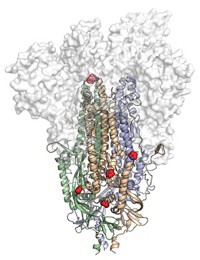
McLellan and his team scoured the structure for clues that could help them stabilize the MERS spike protein. They homed in on a region toward the top of the spike, a small loop of amino acids that held two coil-like structures called α-helices together.
“It is like a spring bent in half,” McLellan says. When the spike protein binds to a human cell, that spring is released, and the two helices and the loop straighten into one long helix that harpoons the human cell and pulls the virus and human membranes close together until they fuse.
Blocking the release of that spring should prevent viral fusion, giving the immune system a chance to make antibodies that prevent infection, they theorized. McLellan’s lab tried several tricks before arriving at one that worked: adding two prolines in the loop between the two helices clamps the spring together. This 2P mutation enabled McLellan and Ward to solve the MERS prefusion spike structure in 2017 (Proc. Natl. Acad. Sci. U.S.A., DOI: 10.1073/pnas.1707304114). Graham began working with Moderna to make an mRNA vaccine for MERS using the 2P mutation that same year.
Once the genetic sequence of SARS-CoV-2 was released this January, Graham’s lab, collaborating with McLellan’s lab, was able to compare its genome with those of SARS and MERS, pinpoint the code corresponding to that bent spring, and then add the 2P mutation to lock the SARS-CoV-2 spike in its prefusion conformation.
Several other companies developing COVID-19 vaccines are also using the 2P mutation. Dan Barouch, a Harvard Medical School vaccine scientist working with J&J on its COVID-19 vaccine, tested six versions of the vaccine in 35 monkeys. The spike protein vaccine with the 2P mutation performed the best. “It looks promising,” says Bing Chen, a virologist at Harvard Medical School who was part of the study. But he cautions that the 2P mutation didn’t make a huge difference, and even if it is the best option, there’s no guarantee it will work as well in humans.

After rapidly determining the structure of the SARS-CoV-2 prefusion stabilized spike protein in February, McLellan’s lab continued to tinker with the spike protein. After testing more than 100 variations, he published a new version in July called HexaPro (Science 2020, DOI: 10.1126/science.abd0826). It contains the original 2P mutation plus four additional prolines that further stabilize other regions of the protein. His preliminary results suggests the molecule might be 10 times as potent as 2P.
Advertisement
“There is probably no limit to how much more we can keep going or how much time we can keep spending,” McLellan says, but he’s happy with HexaPro. The HexaPro gene has been shipped to dozens of labs, and multiple vaccine companies are already conducting lab tests of HexaPro as part of a second-generation COVID-19 vaccine, he adds.
The use of the 2P mutation in coronaviruses is the subject of a patent application filed in 2017 by NIAID, Scripps, and Dartmouth. NIAID and the University of Texas at Austin, where McLellan is now an associate professor, filed another patent early this year. So far, those patent applications are not preventing anyone from using the trick, but the early government investment that made 2P-based vaccines possible could add fuel to debates about how government-funded research should influence vaccine pricing.
It’s too soon to say if Moderna’s vaccine or any of the other front-runners will be successful. Even if they are, the 2P mutation won’t be the only reason, and it might not even be the most important. One of the COVID-19 vaccine front-runners, produced by AstraZeneca, doesn’t even use it.There are countless ways the novel coronavirus has caught us unaware, but if the 2P mutation turns out to be helpful, it may be one of the few preparedness success stories of the pandemic.
“We started quickly because we had an idea of what to do ahead of time,” Graham says. “I think this whole episode is going to change how we think about vaccines going forward.”
Update
This story was updated on Oct. 1, 2020, to clarify that Graham's lab and McLellan's lab were both involved in the early design of Moderna's SARS-CoV-2 vaccine.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter