Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Nuclear Chemistry
Mining medical isotopes from nuclear waste
Actinium-225 is in demand. Nuclear innovation company TerraPower hopes nuclear waste can supply it
by Wudan Yan, special to C&EN
July 27, 2020
| A version of this story appeared in
Volume 98, Issue 29

A half hour north of Seattle in Everett, about 15 black barrels sit in a nondescript storage facility owned by the nuclear innovation company TerraPower. Each barrel is the size of a large trash can, around 110 L, and weighs over 350 kg. What’s held within is . . . complicated. Just inside the thick shell is a layer of foam padding. Within that padding sits a heavy-duty container, which in turn encloses a steel pipe capped on each end. Inside the pipe is a screw-top aluminum can, which holds a thick plastic bag, which encases a glass vial about the size of a tube of toothpaste. “It’s sort of your classic Russian doll,” says Jeff Latkowski, TerraPower’s senior vice president of innovation. That innermost glass vial holds half a gram of a yellow matter—a mixture of the radioactive elements uranium and thorium.
These smidgens of material would normally have been disposed as nuclear waste by the US Department of Energy (DOE) and its partner in disposal, the nuclear waste management company Isotek. Instead, TerraPower requested the samples so that Latkowski and his colleagues can unpack the Russian dolls and extract a valuable medical isotope: actinium-225, which results from radioactive decay of uranium and has shown promise in treating a range of cancers. TerraPower hopes that mining the waste will yield between 200,000 and 600,000 doses of 225Ac a year, 100 times the number of doses currently available globally. TerraPower’s efforts are part of a global push to increase actinium production to ensure a reliable supply for medical research and clinical use.
It’s fortuitous that this medically valuable isotope can be drawn out from nuclear waste that would otherwise have no use, says Jim Bolon, president of Isotek. If any of the ongoing clinical trials using 225Ac to treat cancer pay off, repurposing such waste could be a key source of the element for those treatments.
Actinium’s alpha energy
When 225Ac undergoes radioactive decay, it ejects an α particle composed of two protons and two neutrons. A single α particle targeted at a cancer cell can stop the cell’s growth in its tracks, explains physician and immunotherapy researcher David A. Scheinberg of Memorial Sloan Kettering Cancer Center, who has a number of patents for the medical use of 225Ac and consults for the company Actinium Pharmaceuticals. This whopping punch knocks cancer cells out so completely, he says, that so far little resistance to the treatment has evolved—a huge plus when treating tumors that can quickly morph into drug-resistant forms. What’s more, although α particles pack a lot of energy, they travel only a short distance through the body—about the diameter of two or three human cells—so there’s minimal risk of damage to tissues beyond the target.
In 1993, scientists discovered that 225Ac could be attached to antibodies or other cancer-targeting biomolecules that carry the isotope to the target cells, delivering the radiation precisely where it’s needed. And actinium is an ideal candidate to attach to drugs because it’s a charged metal, Scheinberg explains, which enables it to tightly chelate an antibody.
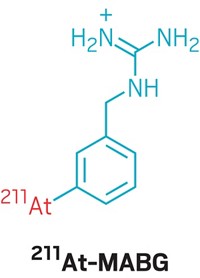
In addition, unlike other short-lived radioisotopes such as bismuth-213, which has a half-life of 45 min, and astatine-211, which has a half-life of 7 h, 225Ac has a relatively long half-life of 10 days. That gives scientists more time to extract it, process it into a drug, and deliver that drug to a patient. Currently, such actinium drugs are being studied in preclinical models of breast cancer and glioblastoma, and clinical trials are ongoing in patients with acute myeloid leukemia and prostate cancer.
No actinium-based drugs are yet approved by the US Food and Drug Administration, but if any get a green light, the medical community will need multiple ways to produce 225Ac and multiple institutions producing it, says Kevin John, project manager for the DOE’s Tri-Lab Effort to produce actinium. Currently, the Oak Ridge National Laboratory (ORNL), Brookhaven National Laboratory (BNL), and Los Alamos National Laboratory (LANL) are the country’s sole providers of its limited supply of 225Ac. In 2018, the International Atomic Energy Agency convened a meeting to discuss a global strategy to meet the rising demand for 225Ac. The resulting report described production via multiple sources, including proton cyclotrons, linear accelerators, and nuclear waste.
Milking the thorium cow

In 2014, Latkowski and radiochemist Ken Czerwinski of TerraPower wanted to take a second look at getting medical isotopes from nuclear waste—an idea attempted at smaller scales in the early 2000s but not pursued further or scaled up because of the lack of infrastructure for extracting actinium. Last fall, however, TerraPower—launched and chaired by Bill Gates—announced a partnership with Isotek to develop such infrastructure. The DOE already contracted Isotek to eliminate the nation’s remaining 233U, originally intended for use in nuclear reactors. 233U is a security concern because it is highly radioactive and is a potential weapon material. The amount of 233U in the US is not publicly available.

By the time Isotek gets the material for disposal, some of the 233U has already decayed to thorium-229, which in turn decays to 225Ac. As part of its disposal process, Isotek dissolves the material in nitric acid to turn the uranium into uranyl nitrate, which is less radioactive and can be buried in a designated site. Isotek runs the solution of uranyl nitrate through an ion exchanger to selectively bind the thorium. It washes the thorium off the column, dries it, and ships it to TerraPower to recover actinium.
TerraPower plans to recover 225Ac from 229Th by a method known as “milking the thorium cow.” Saed Mirzadeh and Rose Boll of ORNL developed the method in the early 1990s, and the lab still uses the method today to produce 225Ac for medical use. ORNL scientists take the thorium extracted from 233U and run it through a column. A resin in the column selectively binds thorium, while the “milk”—actinium and other radioisotopes—flows through. An additional ion-exchange step recovers pure 225Ac from the milk. The amount of 225Ac generated each year from ORNL’s thorium cow is only enough to support very limited clinical trials, Mirzadeh told C&EN. TerraPower will be scaling up this isolation method to process the larger supply of thorium provided from waste—effectively creating an actinium dairy.
Still, the amount of available 225Ac is at the mercy of natural processes that happen over a very long time. The natural decay of 233U and 229Th is steady and reliable but also incredibly slow. 233U has a half-life of 160,000 years, and the half-life of 229Th is 8,000 years. The long half-lives mean that every year, a kilogram of 233U yields only only about 5 mg of 229Th, which can then decay into the medically valuable actinium. From the remaining 233U that Isotek is set to dispose, TerraPower is on track to receive all the thorium that can be extracted during processing, Bolon says. That means TerraPower will get at least 45 g of 229Th a year, enough to provide 50 to 100 times as much 225Ac as the DOE is able to provide.
In 2008, shortages of actinium affected clinical trials to treat acute myeloid leukemia, and researchers warned that if the DOE buried its inventory of 233U, further clinical research using actinium would be threatened: as it stands, only 1 in 5,500 people with cancers who could benefit from actinium therapies are getting that treatment. To address this shortage, the DOE has also invested in efforts to manufacture 225Ac. At LANL and BNL, researchers make actinium using high-energy particle accelerators. They start with a piece of 232Th—an abundant material that is not radioactive—about the size of a thin hockey puck. The accelerator uses a proton beam to bombard the puck, and over the course of days, generates actinium and many other fission products. That mixture of isotopes must be further separated and purified to recover pure 225Ac. The accelerator route can produce actinium on demand, rather than needing to wait 2 months at a time for enough 229Th to naturally decay.
Nevertheless, having more sources for this medical isotope would invariably help advance medical research, says Cathy Cutler, director of medical isotope research and production at BNL. With permits to accept and handle thorium at its Everett site now in place, TerraPower hopes to begin work in the fall on those 15 barrels of thorium that it already has stored. Until then, and with some delays due to the novel coronavirus, it is designing and building the facility, ensuring engineering rigor and implementing shielding so that everything will be done safely, Latkowski says. In addition to the 225Ac work, TerraPower is working on other radiochemistry projects, including developing new types of nuclear reactors to produce green energy.
This work and other efforts around the world can help increase global supply of the precious radioisotope, removing obstacles to testing actinium therapies and, if approved, delivering them to clinics in larger scales, Latkowski says. “That’s where we hope we can help it across the next threshold,” he says, “to where bigger companies that have the serious resources to really get these drugs done see it as something they want to use because the supply is there.”

Wudan Yan is a freelance writer. A version of this story first appeared in ACS Central Science: cenm.ag/actinium.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter