Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Chemical Regulation
US EPA changes PFAS rule after signing
Top Democrat on Senate environment committee asks inspector general to investigate
by Britt E. Erickson
August 6, 2020

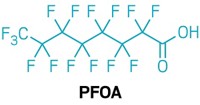
The US Environmental Protection Agency is facing backlash over the way it finalized a rule that requires companies to notify the agency 90 days before making or importing products containing certain long-chain per- and polyfluoroalkyl substances (PFAS). These chemicals, which include perfluorooctanesulfonic acid (PFOS) and perfluorooctanoic acid(PFOA), persist in the environment and are linked to cancer, thyroid disease, immune problems, and decreased fertility.
EPA Administrator Andrew Wheeler signed the rule June 22, but the EPA published a different version of it in the Federal RegisterJuly 27.
The published version omits a requirement that any part of a product coated with certain PFAS is subject to the rule. Instead, the rule says the EPA plans to issue guidance on which coatings are covered by the rule.
The top Democrat on the Senate Environment and Public Works Committee, Sen. Tom Carper, is calling for the EPA’s inspector general to investigate whether the process the EPA used to alter the rule was appropriate and legal.

Carper first raised concerns about the rule in an April letter to the EPA, noting that the rule proposed in March was much weaker than the version first proposed in 2015. Carper claimed to have documents indicating that White House official Nancy Beck spent years trying weaken the rule. The version of the rule signed by Wheeler on June 22 did not include several of the weakening changes.
“However, in recent weeks, my office learned that Dr. Beck continued to push for changes to the rule even after it was signed,” Carper says in a statement. The rule published July 27 “includes significant changes that my office was informed were directed to be included by Dr. Beck.” Beck previously worked as senior director of regulatory affairs at the American Chemistry Council, the US chemical industry’s main lobbying group. She joined the EPA’s chemical safety office in 2017, then moved to the White House National Economic Council in 2019. President Donald J. Trump has nominated Beck to lead the Consumer Product Safety Commission.
“It is not unusual for some minor technical or typographical corrections to be made to a rule after it has been signed but before it is published in the Federal Register,” Carper says. However, when significant errors occur, a rulemaking process is required to correct the rule and notify the public of the changes, he says.
The EPA did not notify the public that significant changes were made to the PFAS rule after it was signed.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter