Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biocatalysis
Light gives enzyme a radical new role
Chemists redeploy reductase enzyme to form lactam rings
by Mark Peplow
June 20, 2019
| A version of this story appeared in
Volume 97, Issue 25

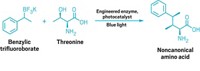
They say you can’t teach an old dog new tricks. But bathing an enzyme in blue-green light has enabled researchers to completely change its catalytic function, so that it carries out a ring-forming reaction not found in nature. The team, led by Todd K. Hyster at Princeton University, used this strategy to build a range of cyclic lactams, a motif found in pharmaceuticals and agrochemicals (Science 2019, DOI: 10.1126/science.aaw1143).
Enzymes are highly efficient catalysts, able to work at room temperature and in water. They can also control the stereochemistry of the molecules they make, selectively producing one isomer over another.
There are still plenty of reactions that natural enzymes cannot tackle, though. Researchers may extend an enzyme’s repertoire by changing its amino acid sequence, or by inserting a transition metal catalyst into its structure, but these approaches are not always successful. So over the past few years, Hyster’s team has developed an alternative way to revamp an enzyme’s activity that relies on light activation.
Their latest effort involves a family of enzymes called ene-reductases, which are thought to help certain microbes combat oxidative stress. The enzyme’s large active site harbors a flavin cofactor that plays an essential role in its natural reaction, transferring a hydride anion and a proton to double bonds to reduce troublesome metabolites.
But Hyster’s team found that shining cyan light onto the flavin puts it into an excited state that is primed for a rather different process. “In its activated state, it is a potent single-electron reductant,” Hyster says. This enables a radical reaction that can transform a variety of α-chloroamides into lactam rings containing 5 to 8 atoms. The reaction can also work at gram-scale, and in the open air.
The team ran a series of experiments to figure out the radical reaction’s mechanism. First, the light-activated flavin delivers an electron that pries chloride from the α-chloroamide, generating a carbon radical. This reacts with a carbon-carbon double bond in another part of the molecule, forming a ring and another carbon radical. Finally, this radical plucks a hydrogen atom from the flavin to produce the finished lactam. Another reducing agent in the reaction mixture regenerates the flavin so that it’s ready for the next target molecule.
Crucially, the α-chloroamide and flavin form a complex within the active site during the reaction, and in many cases this chiral environment ensured that only one of the lactam’s stereoisomers formed. “It’s a reaction that natural enzymes do not catalyze, and most importantly it is stereoselective,” says Uwe T. Bornscheuer at Greifswald University, who works on enzyme catalysis. “It’s a cool paper.”
The researchers mostly worked with a reductase from Gluconobacter oxydans, but for reactions where this enzyme offered poor yields or low stereoselectivity the researchers could usually find another reductase from a different organism that would improve the results.
They also had a stroke of luck when a mutant form of their G. oxydans reductase, in which a single amino acid residue was altered, gave a slightly higher lactam yield than its natural cousin. The discovery was a happy accident: “We actually had a small typo when we ordered the gene” used to make the enzyme, Hyster says sheepishly. “I’ll be honest, that was entirely my mistake.”
Hyster has previously used the same light activation strategy to repurpose other reductases (Nature 2016, DOI: 10.1038/nature20569; Nature Chem. 2018, DOI: 10.1038/s41557-018-0059-y), but this is the first time they have applied it to make carbon-carbon bonds. The researchers now hope to develop their oddball enzyme reactions as tools for pharmaceutical synthesis.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter