Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Catalysis
Chemists simplify aniline synthesis
One-pot photoredox method ditches aromatic chemistry
by Leigh Krietsch Boerner
August 10, 2020

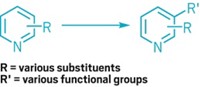
Aromatic rings are important in medicines and agricultural products, but attaching functional groups to them is a persnickety process. Chemists can only add substituents at specific positions on the ring, and this can take multiple steps that require complex ingredients, and give a mixture of products that are hard to separate.
Now chemists from AstraZeneca and Daniele Leonori’s group at the University of Manchester have worked out a one-pot cascade reaction to make anilines—benzene compounds attached to an amino group—without having to mess with finicky aromatic-ring chemistry (Nature 2020, DOI: 10.1038/s41586-020-2539-7).
“Our idea was to go as far away as possible from aromatic chemistry,” Leonori says. His team decided to start with an amine and a cyclohexanone, two molecules that don’t have any carbon–carbon double bonds. Their reaction happens in a single pot at near room temperature, but it goes through several steps to ultimately get the aniline. First, the amine and cyclohexanone go through a coupling reaction to form the C–N bond. Then, with the help of blue light, an iridium photocatalyst pulls an electron off the resulting enamine, generating a radical. A cobalt redox catalyst sequentially grabs hydrogen atoms from the radical’s cyclohexyl ring, ultimately making aromatic aniline (shown). Leonori and coworkers synthesized over 80 aniline compounds using this method, and got yields from 43 to 96%.
Leonori says that they still need to scale up and automate the process, but he hopes it will provide an alternate route to commercially important anilines. For the anilines the group made, the syntheses have fewer steps than the aromatic-based industry standard reactions.
Paolo Melchiorre, an organic chemist at the Institute of Chemical Research of Catalonia, says that the new method is simple and may “quickly find widespread application in chemical synthesis.” Nan Zheng, an organic chemist at the University of Arkansas, calls the reaction an elegant solution to a longstanding problem. “This is another excellent piece of work in photoredox catalysis to push the boundaries of organic chemistry,” he says.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter