Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Catalysis
Nickel catalyst fends off air attack
Air-stable complex offers a more convenient approach to coupling reactions
by Mark Peplow
December 9, 2019

Nickel catalysts have shot to synthetic stardom over the past decade or so, proving particularly useful in coupling reactions that form carbon-carbon or carbon-nitrogen bonds. The most popular catalyst for these reactions is bis(1,5-cyclooctadiene)nickel (Ni(COD)2), and although it’s very effective, the complex decomposes rapidly in air, making it troublesome to use.
Researchers at the Max Planck Institute for Coal Research have now developed an air-stable alternative to Ni(COD)2 that could make nickel catalysis much more accessible to users (Nat. Catal. 2019, DOI: 10.1038/s41929-019-0392-6). “Air stability has been a huge challenge,” says Josep Cornellà, who led the research, “and a lot of people don’t have access to glove boxes.”
Nickel is increasingly used in reactions traditionally dominated by palladium catalysts. Not only is nickel cheaper, Cornellà says, it can also catalyze a wider range of reactions than palladium. But the nickel catalysts used in these reactions are often extremely reactive, so they must be freshly generated within the reaction mixture. Chemists do this by mixing a precatalyst like Ni(COD)2 with other ligands, such as phosphines or bipyridines, which take the place of cyclooctadiene to form the active catalyst.

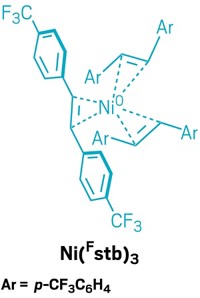
The new precatalyst is based on a nickel atom surrounded by three stilbene ligands that bear trifluoromethyl groups (Ni(Fstb)3). These ligands enclose the nickel and protect it from oxygen. Cornellà’s team could make the red solid in 20 g batches by mixing a cheap precursor—nickel acetylacetonate—with the stilbene ligand and a reducing agent called triethylaluminum. The complex is stable for months in a freezer and only starts to decompose after several days at room temperature.
“People will use this immediately because it’s a drop-in replacement,” for Ni(COD)2, says Nilay Hazari of Yale University, who develops transition metal catalysts. “In my mind, it’s a very significant step forward.”
The Max Planck researchers used Ni(Fstb)3 to generate active catalysts in more than a dozen different reactions, including classics such as a Suzuki-Miyaura coupling, Buchwald-Hartwig carbon-nitrogen bond formations, and a Heck reaction. It generally performed as well as Ni(COD)2, giving the intended products in high yields.
Teamed with a carbene ligand, the new complex also catalyzed a coupling reaction between pentafluorobenzene and an alkyne, a feat that Ni(COD)2 cannot manage. Some reactions were less successful, though—a coupling reaction that involved butadiene or isoprene failed because these compounds could not displace the stilbene ligands from the complex.
Hazari says that Ni(Fstb)3 could be particularly helpful in catalyst development studies. For example, it could easily be loaded onto a 96-well plate with a different ligand in each well to assess which of the resulting active catalysts were most effective for a particular reaction. “For screening ligands, this is fantastic,” Hazari says.
However, he thinks there is still room for improvement. Triethylaluminum can ignite spontaneously in air, so it would be better to use a safer reducing agent to prepare the complex. It would also be helpful to improve the complex’s thermal stability, he adds. Cornellà and his colleagues are now testing a library of other stilbene ligands to fine-tune the reactivity of their complex.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter