Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Catalysis
Powerful acid prevails upon reluctant alkenes to react
Unactivated alkenes yield to Brønsted acid catalyst to form new chiral C–O centers
by Tien Nguyen
April 2, 2018
| A version of this story appeared in
Volume 96, Issue 14

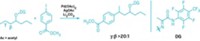
To drive reactions forward, catalytic Brønsted acids wield protons to provoke molecular bonds into action. These organic catalysts have acted on obliging, heteroatom-containing bonds in a variety of transformations, but they’ve been unable to tempt simple, unactivated alkenes—a class of substrates that has been dominated by efficient transition-metal catalysis. Researchers led by Benjamin List at the Max Planck Institute for Kohlenforschung have now breached this barrier in reaction scope for Brønsted acids using a highly acidic, chiral imidodiphosphorimidate (IDPi) compound (Science 2018, DOI: 10.1126/science.aaq0445). The researchers propose that the substrates—1,1-disubstituted alkenes with pendant alcohols—squeeze into an enzymelike pocket in the catalyst. The stereodefined cavity facilitates a ring-forming reaction to give tetrahydrofuran and tetrahydropyran products in high enantioselectivity. The team additionally reveals one example of an intermolecular version of the reaction, albeit with slightly lower yield and selectivity than its tethered counterpart. This transformation could be the first of many such alkene hydrofunctionalizations, List says.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter