Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Catalysis
Straightforward path to linear alkylbenzenes identified
Nickel catalyst provides a route to the cleaning product chemicals with high yields and selectivity
by Leigh Krietsch Boerner
February 10, 2020

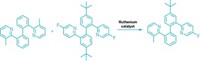
The world used about 3.5 million metric tons of linear alkylbenzenes, worth about $4.8 billion, in 2019, mostly to make detergents and cleaning products. However, the industrial synthesis of these compounds tends to make mixtures containing branched alkylbenzenes, which don’t biodegrade easily and can pollute rivers, lakes, and oceans. Anti-Markovnikov transition metal–catalyzed hydroarylation, in which the aryl compound bonds to the less substituted carbon of an alkene, could make linear alkylbenzenes, but these reactions have been plagued by poor yields and selectivity.
Now John Hartwig of the University of California, Berkeley; Yoshiaki Nakao of Kyoto University; and coworkers have coupled select arenes with an unconjugated terminal alkene to form linear alkylbenzenes in one step with 85–96% yields and high selectivity (Nat. Chem. 2020, DOI: 10.1038/s41557-019-0409-4).
The current acylation and reduction methods to produce these compounds is through a Friedel-Crafts acylation, which requires multiple steps. The new reaction is the first to be done in one step, with approximately 50:1 linear to branched selectivity, Hartwig says. In addition, the catalyst, a nickel cycloocta-1,5-diene compound, could turn over 280 times and still give an 85% yield. This turnover is more than 10-fold higher than reactions involving catalytic hydroarylation of benzene, Hartwig says.
Normally in these reactions, the alkenes isomerize between internal and terminal alkenes, Hartwig says. If the reaction is not selective for terminal alkenes, it will either give a mixture of linear and branched alkylbenzenes, or more or less stop. This new reaction only occurs with the terminal alkene, which gets around the isomerization problem. The researchers didn’t intentionally design this selectivity into their system, but it’s a pathway that in principle could be generalized to similar types of X–H bonds adding to alkenes, Hartwig says.
Hartwig and coworkers found that the reaction goes through an alkyl nickel–aryl intermediate and involves an unexpected hydrogen transfer between catalyst ligands followed by reductive elimination. Further analysis of the mechanism revealed that the bulk of the N-heterocyclic carbene ligands did not affect the activity. The reactivity has more to do with noncovalent attractive interactions between ligands, instead of repulsive ones, Hartwig says.
These high-selectivity results from Hartwig and coworkers achieve a long-standing goal, says T. Brent Gunnoe, an organometallic chemist at the University of Virginia. “The use of large N-heterocyclic carbene ligands and noncovalent interactions to enhance the rate of catalysis is an unexpected result and a clever idea that could have broader impact in the design of other catalysts,” he says.
If the catalyst activity were high enough, this reaction would be easy to scale up to the industrial level, Hartwig says, because it’s a simple addition reaction, and there are no side products. However, it’s not perfect. “The biggest drawback of this system, in addition to catalyst lifetime, is its low tolerance for a lot of functional groups,” such as esters and nitriles, he says.
UPDATE
This story was updated on Feb. 12, 2020, to include the name of a corresponding author, Yoshiaki Nakao of Kyoto University.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter