Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Medicinal Chemistry
New reaction creates elusive N-trifluoromethyl carbonyl compounds
Silver fluoride catalyst is key for making these medicinally relevant molecules
by Bethany Halford
September 5, 2019
| A version of this story appeared in
Volume 97, Issue 35

There’s a silver lining for chemists trying to make N-trifluoromethyl carbonyl compounds. Using silver fluoride as a key reagent, a team at RWTH Aachen University has developed a general method for making these elusive compounds, including N-trifluoromethyl amides, carbamates, thiocarbamates, and ureas (Nature 2019, DOI: 10.1038/s41586-019-1518-3).
The reaction could help medicinal chemists on the hunt for new drugs. These chemists often incorporate amides into their drug candidates because the motif can help a compound bind to its biological target. As a result, amides show up frequently in best-selling small-molecule drugs. But amides are also sites where the body’s enzymes can metabolize drug molecules. If chemists could add a trifluoromethyl group—which tends to resist biological breakdown—to an amide’s nitrogen, they might be able to make a drug last longer in the body.
Advertisement
Until now, making these N-trifluoromethyl amides has been tricky. Such reactions required toxic or extremely reactive reagents, like BrF3. Also, the reactions couldn’t be performed on compounds containing reactive functional groups or stereocenters, which are common in pharmaceuticals.
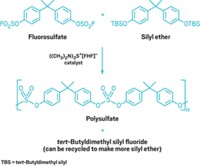
The new synthetic method, developed by Franziska Schoenebeck, Thomas Scattolin, and Samir Bouayad-Gervais, uses AgF to combine an isothiocyanate with bis(trichloromethyl) carbonate. The AgF plucks the sulfur from the isothiocyanate and adds three fluorines to its carbon atom. Another AgF-catalyzed reaction with bis(trichloromethyl) carbonate forms an N-trifluoromethyl carbamoyl fluoride. Subsequent reaction of these stable N-trifluoromethyl carbonyl compounds with nucleophiles forms a range of complex N-trifluoromethyl carbonyl compounds (example shown).
“The clever combination of reagents and diverse range of targets makes this an appealing route,” says Jonathan Clayden, an expert in organic synthesis at the University of Bristol.
The challenge, he says, comes in scaling up the reaction should a drug candidate made in this manner make it to clinical trials. The large excess of silver salts used would need to be recycled efficiently. Schoenebeck points out that silver metal has a high recycling rate, which might be applied to the by-products of this reaction.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter