Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Reagents
Testing how much trisubstituted amines can bulk up
Using a multitude of methods, chemists make unprecedented crowded alkyl amines; but one elusive target remains out of reach
by Tien Nguyen
May 4, 2018
| A version of this story appeared in
Volume 96, Issue 19
By nature, nitrogen prefers to bond with three molecular companions. But, when it comes to bulky alkyl groups, amines are choosy about the company they keep and have resisted many chemists’ campaigns to form highly hindered tertiary amines. This class of alkylamines, though, has been useful in many applications, including as precursors to photostabilizers and polymerization inhibitors and as reagents in catalysis, since their bulk blocks them from binding and deactivating catalysts.
Now a team led by Klaus Banert at Chemnitz University of Technology has evaluated 10 previously reported approaches to synthesize more than a dozen alkylamines that the researchers claim have unprecedented steric crowding (J. Org. Chem. 2018, DOI: 10.1021/acs.joc.8b00496).
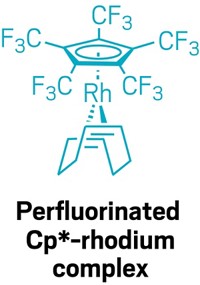
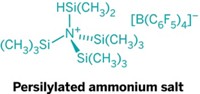
The researchers observed that the main obstacle to making the hindered amines was their tendency to undergo Hofmann-like elimination reactions. In this type of reaction, the nitrogen plucks a hydrogen off a neighboring carbon to kick off one of the alkyl group substituents, leaving behind a less bulky disubstituted amine. The group proposed that while tert-butyl substituted amines were particularly susceptible to this degradation pathway, the arguably larger adamantyl groups would be immune to such elimination given the orientation of its pluckable hydrogens.
Despite numerous attempts, the authors were unable to make tri-tert-butylamine, one of the most hindered of tertiary amines. Banert and his colleagues question whether anyone will ever isolate the compound at room temperature. Chemists have long debated this idea.
Twenty-two years ago, Armin de Meijere, professor emeritus at University of Göttingen, and colleagues made di-tert-butylcyclopropylamine, previously thought to be one of the most hindered tertiary amines isolable at room temperature. He thinks that the new work pushes the limits of sterically congested amines forward considerably, but agrees that tri-tert-butylamine will probably never be made.
Peter Livant, who is now retired from Auburn University, thinks tri-tert-butylamine may one day show itself, but whether it’s stable for one second or 20 years is unknown. “What struck me about this article is that they were after a sort of Holy Grail. They wanted to reach the top of a mountain that’s never been scaled before,” Livant says. “That sense of adventure was really nice.”



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter