Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Analytical Chemistry
Platinum From Breast Implants
Study implicates silicone implants as source of harmful platinum species
by Mitch Jacoby
April 17, 2006
| A version of this story appeared in
Volume 84, Issue 16

Women with silicone breast implants are likely to have higher concentrations of harmful forms of platinum in their bodies than women who have never had implants, according to a new study that is being contested by specialists in silicone chemistry (Anal. Chem., published online April 1, dx.doi.org/10.1021/ac0514016). A key finding of the investigation, which probed the levels and oxidation states of the metal, is that platinum species remain in the body long after the implants have been removed.
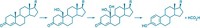
One of the steps in preparing medical-grade silicone (polydimethylsiloxane) is cross-linking the polymer chains to control the material's stiffness and viscosity. The process calls for a cross-linking catalyst such as hexachloroplatinate, which is implicated in the study as the source of the metal.
The study's authors note that exposure to platinum species with oxidation states other than zero is believed to be harmful to human health. In particular, exposure to platinum salts has been associated with neurotoxicity, carcinogenicity, and other harmful effects, they say.
To assess the concentrations of platinum, Ernest D. Lykissa, a forensic toxicologist at ExperTox, in Deer Park, Texas, and Susan V. M. Maharaj of the Center for Research on Environmental Medicine, in New Market, Md., analyzed samples of tissues and body fluids from some 20 subjects with breast implants by ion chromatography-inductively coupled plasma mass spectrometry. The subjects included women of various ages who had had the implants for as long as 25 years. Most of the subjects had had the implants removed-some shortly before the study and others several years before. Control subjects who never had breast implants were also studied.
On the basis of the investigation, the team concluded that platinum concentrations in breast-implant subjects are much higher than in women with no known exposure to platinum. In urine, for example, the researchers found an average of approximately 2 µg of platinum per gram of creatinine, which is 60 to more than 1,700 times higher than in the general population. In breast milk, hair, and nails, they found the platinum concentrations to be higher by factors of 100, 14, and three, respectively. In implant subjects, platinum was found primarily in reactive forms, including +2, +4, and +6 oxidation states, they add.
Silicone experts question the study's findings. For example, Thomas H. Lane, a senior researcher at Dow Corning, which manufactured silicone breast implants until 1992, states emphatically that the material upon which the study is predicated, a hexachloroplatinate (in which platinum is in the +4 oxidation state), was not used as the cross-linking catalyst. Instead, the manufacturer used a vinyl-complexed platinum compound, which enters and exits the catalytic cycle in the Pt(0) oxidation state, Lane says. He also notes that the study contradicts earlier investigations of cadavers that found no statistical difference in the amount of platinum in women with and without implants.
Lykissa acknowledges that the fresh catalyst is in a vinylcomplexed Pt(0) form but contends that years in the body expose the implants to heat and stress that lead to deterioration and oxidation.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter