Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Natural Products
What’s that Stuff
What are frankincense and myrrh and why is their smell so mystical?
Culture and chemistry meet in fragrant plant-based incense
by Carmen Drahl
December 22, 2008
| A version of this story appeared in
Volume 86, Issue 51
SPICY-SMELLING frankincense and myrrh have been intimately intertwined with humanity throughout recorded history, from the frankincense pellets found in the ancient tomb of Pharaoh Tutankhamun to the myrrh-infused brandy concoction used to preserve the body of Vice Admiral Horatio Nelson, a 19th-century British war hero. The substances' musky plumes of smoke are most often associated with embalming, perfumes, and religious rituals around the world, including ceremonies in the ancient temples of Jerusalem and modern Roman Catholic liturgies. Beyond those uses, frankincense and myrrh may also have medicinal and psychoactive components.
Both of the earthy entities are gum resins, which are viscous secretions from trees. Frankincense, also known as olibanum, comes from select trees in the Boswellia genus, and myrrh usually comes from Commiphora trees. The plants belong to the same botanical family and commonly grow on the Arabian Peninsula, in India, and in northeastern Africa. "Both trees are usually gnarled and look stunted, without very many leaves," describes Kerry Hughes, an ethnobotanist and founder of EthnoPharm, a consulting company specializing in plant products.
To access the aromatic resins, locals slice gashes into frankincense and myrrh trees at harvest times and collect the milky resins that ooze from their bark, Hughes says. Once exposed to air and sun, myrrh dries and hardens to reddish-brown pea-sized chunks, whereas frankincense dries to pale yellow, tear-shaped droplets about half that size.
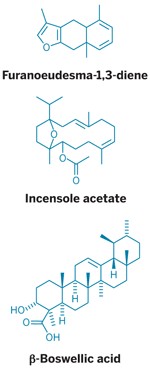
Because factors such as geography and climate affect plant biochemistry, it's impossible to precisely pin down an exact molecular composition for myrrh or frankincense. However, the resins do contain sugar chains, proteins, and steroids, and are mostly a blend of terpenes, a diverse family of hydrocarbons made from five-carbon building blocks. For example, frankincense contains five-ringed triterpenoids called boswellic acids, as well as an array of mono- and sesquiterpenes that contribute to its scent, including α- and β-pinene, limonene, and 4-terpineol. Myrrh's aroma, meanwhile, comes mostly from furanosesquiterpenes such as furanoeudesma-1,3-diene.
Humans have been investigating frankincense and myrrh for centuries, says Arieh Moussaieff, a postdoctoral fellow in plant sciences at the Weizmann Institute of Science, in Rehovot, Israel. "The early scientists did not have the chemical and analytical tools that we have today," so they conducted more straightforward experiments such as dividing the resin into water-soluble and fat-soluble fractions and then looking for desirable bioactivities, he says. Despite today's vast improvements in instrumentation, the biological effects of these resins still aren't fully understood.
For instance, the resins' reputed pain-relieving properties are an active research topic. A study led by an expert at the University of California, Davis, suggested that a frankincense extract provides relief to patients with arthritic knees (Arthritis Res. Ther. 2008, DOI: 10.1186/ar2461). Such extracts have been used for centuries in traditional medicine to treat a range of conditions. However, the new study was funded by a company that makes frankincense extract, and the claims have not been evaluated by the Food & Drug Administration.
In another study, researchers at the University of Florence, in Italy, set mice on a hot plate and compared how long it took regular mice and mice fed myrrh to lick their paws, a sign that the heat was causing them pain. Sure enough, mice dosed with myrrh held out longer. The team's additional tests suggested that furanoeudesma-1,3-diene and another terpene are primarily responsible for the analgesic effects and that the compounds affect opioid receptors in the mice's brains, which influence pain perception (Nature 1996, DOI: 10.1038/379029a0). The precise pathways these compounds affect have yet to be determined, however.
Frankincense also affects mouse brains, and in a way that provokes fascinating questions about the intersection of culture and chemistry. "Most present-day worshippers assume that incense burning has only a symbolic meaning," Moussaieff says. But together with his Ph.D. adviser Raphael Mechoulam of the Hebrew University of Jerusalem and an international team of coworkers, he found evidence that a compound in frankincense resin exhibits depression- and anxiety-dampening effects in mice (FASEB J. 2008, DOI: 10.1096/fj.07-101865)). The team also demonstrated that the compound, a diterpenoid called incensole acetate, activates an ion channel involved in warmth perception in the skin.
Although the results haven't been confirmed in humans, "it is possible that incensole acetate augments the euphoric feeling produced during religious functions," Moussaieff notes. Given that incense is one of the common threads in most major world religions and that immense symbolism is attached to incense burning, the Israeli team's findings "don't surprise me at all," Hughes says.
UPDATE: This article was modified on Dec. 19 2016 to refresh its information and data.





Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter