Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Silver Bullet For Fluorinations
ACS Meeting News: Late-stage cross-coupling may open route to radiotracers
by Bethany Halford
August 24, 2010
| A version of this story appeared in
Volume 88, Issue 35

Banking on the chemistry of silver, scientists have developed a new cross-coupling reaction that allows them to tack fluorine atoms onto aromatic substituents, even in densely functionalized molecules. The reaction, which was presented today at the American Chemical Society national meeting in Boston, could be a boon to the synthesis of radiotracers for positron emission tomography (PET), for which new methods to install 18F during the final stages of synthesis are needed.
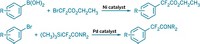
The reaction, developed by Tobias Ritter, Pingping Tang, and Takeru Furuya of Harvard University, uses silver oxide to catalyze the fluorination of aryl tin compounds with the electrophilic fluorinating reagent N-chloromethyl-N-fluorotriethylenediammonium hexafluorophosphate (J. Am. Chem. Soc., DOI: 10.1021/ja105834t). The reaction "is the first example of silver catalysis for carbon-heteroatom bond formation by cross-coupling chemistry," Ritter notes.
"One area my group is terribly interested in is developing new ways to develop tracers for PET imaging," Ritter says. Such syntheses dictate that short-lived radioactive isotopes are incorporated during the final steps of making the molecule. "PET with 18F is currently limited by the absence of general chemistry that can introduce fluorine into molecules at a late stage," he explains. He believes the new reaction could help solve that problem.
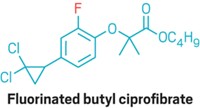
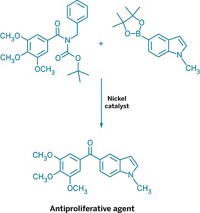
Ritter's group demonstrates the versatility of its new cross-coupling by using it to fluorinate polypeptides, polyketides, and alkaloids. They show that many functional groups, including a vinyl ether, a dienone, alcohols, an allylic alcohol, ethers, esters, and an oxetane survive the reaction unscathed. "To date, no other fluorination reaction has been shown to have a substrate scope as broad as that shown here," Ritter points out.
"Late fluorination processes for highly functionalized molecules are in high demand, and the silver-catalyzed carbon-fluorine bond formation developed by Ritter is a great advance," comments Véronique Gouverneur, a chemistry professor at Oxford University, in England, who recently developed 18F-labeled Selectfluor (Angew. Chem. Int. Ed., DOI: 10.1002/anie.201002310), a fluorinating reagent that's very similar to the one Ritter's group uses. "This work is certain to find immediate application in pharmaceutical research and beyond," she says.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter