Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Zinc Dianion Drives Cross-Coupling
An organozinc dianion, not a monoanion as previously thought, is the active transmetalating reagent in popular organic reaction
by Stephen K. Ritter
June 18, 2012
| A version of this story appeared in
Volume 90, Issue 25
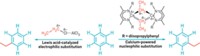
In a surprise finding, Canadian chemists have discovered that an organozinc dianion is the active transmetalating reagent in the Nobel Prize-winning Negishi cross-coupling reaction, not a monoanion species as previously believed (Angew. Chem. Int. Ed., DOI: 10.1002/anie.201203547). The zinc species in a Negishi reaction, typically prepared from an organolithium reagent and a zinc halide, transfers its organo group to a palladium catalyst, which in turn mediates C–C coupling with an organo group contributed by an organohalide. Organic chemists had taken for granted that RZnX2– was the active transmetalator, where R is the organo group and X is a halide. But a team led by Michael G. Organ of York University, in Toronto, and Jason A. C. Clyburne of Saint Mary’s University, in Halifax, Nova Scotia, discovered otherwise. Through titration studies they observed that cross-coupling doesn’t occur when one equivalent of organolithium reagent is used—the organolithium goes to form RZnX2–. Only when additional lithium reagent is added and RZnX32– forms does cross-coupling take place. The team proved the dianion’s pivotal role by synthesizing a (CH3CH2)ZnBr32– salt and showing that cross-coupling proceeds when using it without adding any auxiliary reagents.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter