Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Materials
A Cheaper-Than-Platinum Catalyst
Pollution Control: Oxide material outperforms platinum in converting NO to NO2
by Elizabeth K. Wilson
August 20, 2012
| A version of this story appeared in
Volume 90, Issue 34

In the continuing search for cheaper, more efficient catalysts for cleansing diesel engine exhaust, researchers report a new class of mixed-phase oxides that under laboratory conditions exceed the performance of expensive commercial platinum-based catalysts (Science, DOI: 10.1126/science.1225091).
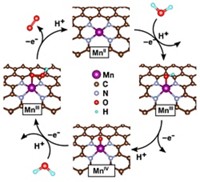
A team of scientists from the U.S., China, and South Korea, led by materials scientists Kyeongjae Cho and Xianghong Hao at Nanostellar Inc. in Redwood City, Calif., report that Mn-mullite(Sm, Gd)Mn2O5—manganese-mullite materials containing either samarium or gadolinium—converts the toxic diesel engine exhaust product nitric oxide to the more benign nitrous oxide.
The researchers investigated the catalyst’s mechanism using infrared Fourier transform spectroscopy as well as density functional theory calculations. They found that its catalytic activity is localized at Mn–Mn dimers on the rough, defect-riddled, or “stepped” mullite surface.
James E. Parks II, who leads an emissions and catalysis research group at Oak Ridge National Laboratory, says the work “shows the benefits of using theoretical simulations to better understand the catalytic processes occurring on new materials.”
Researchers have put a lot of effort into the search for metal-oxide catalysts. For example, scientists reported the development of a strontium-doped perovskite oxide catalyst that outperforms platinum catalysts (C&EN, March 29, 2010, page 11).
However, various factors, including lack of thermal stability, have bedeviled efforts to industrialize them.
Chang H. Kim of General Motors Global R&D, whose team developed the perovskite catalyst, notes the new catalyst’s good NO-to-NO2 conversion abilities but cautions that like other potential catalysts, this material will have to withstand the rigors of real-world conditions.
Yasutake Teraoka, a materials science professor at Japan’s Kyushu University, praised the research. “The development of nonplatinum catalysts for NO oxidation is very challenging,” he says.
Parks also points out that the catalyst might find use in so-called lean-burn engines, which use much less fuel than traditional internal combustion engines. Emissions control systems in these engines are costly and limit their commercialization, he says. The new work, he notes, “may provide a solution for cost-effective lean gasoline emission control.”


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter