Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Materials
Germanes Achieve A Growth Spurt
ACS Meeting News: Chemists devise synthesis of longer chain heavy analogs of alkanes
by Stephen K. Ritter
September 13, 2013
| A version of this story appeared in
Volume 91, Issue 37

The periodic nature of the elements foretells that silicon, germanium, and tin in group 14 should behave like carbon positioned above them and form linear chain molecules. Chemists have had success in making polymeric materials from silicon, germanium, and tin and oligomers of silicon and tin as short-chain models for the polymers. However, they have had limited luck in developing a suitable high-yield synthesis of germanium oligomers.

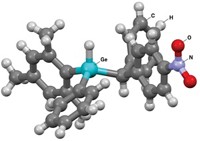
The promise of new optical and electronic properties of the germanium compounds has prompted one research team to keep trying. Kimberly D. Roewe and Charles S. Weinert of Oklahoma State University and coworkers have now reported a synthetic method for making linear germanes, including a hexagermane compound—the longest linear molecular germane characterized to date. Roewe and Weinert presented their work in talks last week in the Division of Inorganic Chemistry at the American Chemical Society national meeting in Indianapolis.
The researchers started by ring-opening a cyclic phenyl-substituted tetragermane they had synthesized previously and adding hydrogen to the ends of the Ge4 chain. They subsequently treated the tetragermane with an isopropylgermanium amide, which enabled them to attach a germanium unit to each end of the chain to form the Ge6 molecule (Chem. Commun. 2013, DOI: 10.1039/c3cc45450a).
“This is an important accomplishment,” said organometallic chemist Lawrence R. Sita of the University of Maryland, College Park. Germanium lies at an interesting part of the periodic table, Sita said. It is not quite a formal nonmetal like silicon or a metal like tin. “Chemists a generation ago first explored the related silicon and tin chemistry but left off without nailing down the germanium analogs,” Sita added.
The Ge6 compound is the first germane long enough to exhibit luminescence, Weinert said. But more dramatic, he noted, is Ge6’s dichroism. When polarized light is shined on the colorless material in one direction, the crystals appear off-white. But when polarized light is shined from a different direction, the crystals exhibit a striking blue color. The way the molecules stack together imparts a type of chirality responsible for the color shift, Weinert suggested. This property could allow the material to be used as an optical filter, he said.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter