Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
Breaking The Ubiquitin Code
Biochemistry: Polyubiquitinated protein synthesis could be used to reveal effects on biology, pathology
by Stu Borman
September 20, 2013
| A version of this story appeared in
Volume 91, Issue 38

Small proteins called ubiquitins attach individually or in chains to other proteins to mark them for disposal, initiate cell signals, or facilitate protein interactions. Three researchers shared the 2004 Nobel Prize in Chemistry for discovering ubiquitination—the process of protein-ubiquitin conjugation—and its role in protein degradation.
Polyubiquitination, in which ubiquitin chains attach to proteins, is often required for ubiquitin-induced biological activity. But preparing homogeneous polyubiquitinated proteins to study the process systematically has been impossible. Despite decades of effort, polyubiquitination’s biological role at a molecular level largely remains a mystery.
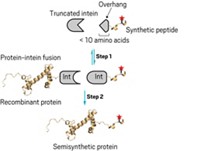
A research team including one of the Nobel Laureates has now synthesized the first di- and tetraubiquitinated proteins in high purity and workable quantities (Proc. Natl. Acad. Sci. USA 2013, DOI: 10.1073/pnas.1315654110). The synthesis has enabled the researchers to evaluate the functional role of polyubiquitination on the protein α-synuclein, which has been linked to Parkinson’s disease.
A nearly endless variety of polyubiquitinated proteins is possible: At least eight types of polyubiquitin chains differing in internal connectivity exist, and they may have different lengths and attach to one of several amino acid residues on the protein. Therefore, a ubiquitination “code”—a system in which specific ubiquitin chains attach to different proteins to produce particular cell signals—may exist. But scientists don’t yet know how to read the code.
Free polyubiquitin chains (unattached to proteins) and monoubiquitinated proteins have been prepared before. Di- and tetraubiquitin chains have also been attached to proteins enzymatically. But the enzymatic process is hard to control and results in heterogeneous mixtures of polyubiquitinated proteins. Pure polyubiquitinated proteins can be isolated from such mixtures, but only in tiny amounts impractical for functional studies.
In the new work, Nobel Laureate Aaron Ciechanover of Technion—Israel Institute of Technology; Hilal A. Lashuel of Swiss Federal Polytechnic Institute of Technology, Lausanne; Ashraf Brik of Israel’s Ben-Gurion University of the Negev; and coworkers used a multistep approach to prepare homogeneous polyubiquitinated α-synucleins. The team used thioester-based linkage reactions to prepare two diubiquitins followed by chemical protein ligation to attach one or both to an α-synuclein modified with a thiolysine. The researchers then desulfurized the thiolysine to form the di- or tetraubiquitinated protein. They used the conjugates to find how ubiquitin chain length affects α-synuclein stability, aggregation, and phosphorylation.
“This is the first time that a target protein has been site-specifically modified with defined ubiquitin chains,” says Eric R. Strieter of the University of Wisconsin, Madison, a specialist in ubiquitination. “It’s a significant accomplishment—something people in the ubiquitin field have been trying to do for a very, very long time.” Nevertheless, the approach is extremely challenging to carry out, Strieter adds. “Additional synthetic advances are still needed in this area, for sure.”
“This is among the largest natural molecules ever synthesized in a lab,” adds protein chemist T. Ashton Cropp of Virginia Commonwealth University. “There is no end to the structure-function experiments that can now be performed to give a better picture of what happens when proteins are modified by polyubiquitins.”




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter