Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Diagnostics
Low-Cost Device Quickly Isolates Blood Plasma
Medical Diagnostics: A plastic device allows researchers to separate plasma from whole blood for measuring HIV viral loads
by Sarah Webb
October 15, 2013

When testing for some infectious diseases, doctors need to look for viruses lurking in blood plasma. In a new study, researchers report a 1.5-inch-tall filtration device that separates plasma from blood cells at larger sample volumes than other low-cost devices can handle (Anal. Chem. 2013, DOI: 10.1021/ac402459h). The filter system could serve as a component of a point-of-care device for monitoring viral loads in HIV patients, the researchers say.
Blood cells and blood proteins can gum up techniques like polymerase chain reaction that doctors use to measure viral loads. Doctors and technicians get around the problem by separating out the straw-colored, watery plasma from blood samples. In a laboratory setting, this is typically done using a centrifuge, a relatively expensive instrument requiring electricity that might not be available in remote areas, particularly in the developing world.
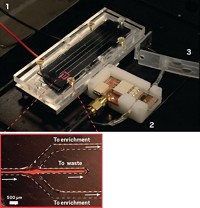
Researchers have tried to remove the cellular components of blood using size-exclusion membranes. Previous designs separate only small volumes of plasma—tens of microliters of plasma from several hundred microliters of whole blood. Blood cells can accumulate on top of the devices’ membranes, blocking the pores and limiting the amount of plasma and viral particles that can flow through, says Changchun Liu of the University of Pennsylvania.
Liu and his team came up with a new design for a plasma filter that leverages gravity to avoid clogging. The rectangular device is separated into two vertical halves by a size-exclusion membrane. The researchers add whole blood to one side of the plastic device, and cells settle to the bottom. Unimpeded by blood cells, plasma and viral particles then pass through the vertical membrane to the other side of the chamber. After five minutes, the researchers use a pipette to remove the collected plasma.
The researchers tested their filtration system on whole blood samples spiked with HIV. They could filter 1.8 mL of whole blood to yield 275 µL of plasma. The team then transferred the separated plasma to a microfluidic chip that they had previously developed to analyze viral loads using a version of polymerase chain reaction.

The ability to separate larger volumes of plasma boosted the sensitivity of the test, Liu says. The researchers could detect viral loads across a wide range of concentrations, from about 350 copies per mL of whole blood to about 35,000 copies per mL.
The device is innovative in its simplicity, says Alexandra Homsy of Swiss Federal Institute of Technology (ETH), Lausanne. She thinks the team should next connect the filter system directly to the detection chip to avoid the need to transfer the plasma. Liu’s group is already working on an integrated device.
Solomon Arman Nabatiyan, founder and chief executive officer of Cervia Diagnostic Innovations, in Evanston, Ill., agrees the new technique represents progress in the field. But he doesn’t think that this particular technique will solve an immediate problem in the developing world. It requires collecting milliliters of blood rather than a finger prick, so using the technique would require trained medical staff to draw blood. Clinics that employ skilled technicians and have refrigeration to store PCR reagents for the microfluidic chip would probably also have the resources to purchase and maintain a centrifuge, he says.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter