Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Analytical Chemistry
Peeking Inside A Capacitor
Energy Storage: Imaging method tracks ion dynamics in real time
by Mitch Jacoby
August 8, 2014
| A version of this story appeared in
Volume 92, Issue 32

A magnetic resonance imaging method for peering inside capacitors while they are charging and discharging is allowing researchers to scrutinize the ion dynamics that govern the operation and performance of these common electrical energy storage devices. The advance may lead to better understanding of the underlying charge-storage mechanism and eventually to improved capacitors.
Similar to batteries, capacitors store and deliver electrical energy. They typically store much less energy than a battery but deliver it on a much faster timescale. Capacitors are an important component of consumer electronics, electric vehicles, and other devices.
Various electrode materials, including nanoporous carbon, can enhance performance relative to capacitors made from other materials. Scientists have proposed various explanations, but because of difficulties in probing the movements of ions in working capacitors, the basis for the enhancement remains unclear.
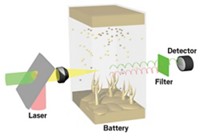
Now, a team led by Clare P. Grey of Cambridge University and Stony Brook University, SUNY, and Alexej Jerschow of New York University has demonstrated that an MRI method can reveal elementary ion processes in a working capacitor (Nat. Commun. 2014, DOI: 10.1038/ncomms5536). The team showed that the technique can monitor—in real time and with high spatial resolution—the rapidly changing location and distribution of anions and cations as they move in and out of the pores of the electrode material and through the electrolyte solution.
The team applied the method, which builds upon Grey and coworkers’ NMR spectroscopy work (J. Am. Chem. Soc. 2013, DOI: 10.1021/ja410287s), to a device with porous carbon electrodes and a tetraethylammonium tetrafluoroborate electrolyte solution.
The group explains that unlike earlier studies, the new method, which measures 1H and 11B MRI signals, monitors ion processes at each electrode simultaneously. Also, their method quickly captures snapshots of the system in nonequilibrium states during electrochemical cycling.
By providing insights into fundamental processes that occur during charging and discharging, this work helps scientists understand the relevant mechanisms of electrochemical processes, says Drexel University’s Yury Gogotsi. He says the method can help identify degradation mechanisms in poorly performing capacitors and might accelerate the development of new and improved materials and devices.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter