Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biobased Chemicals
Making Rubber From Renewables
Supply constraints and high prices give a bounce to raw materials derived from sugar
by Melody M. Bomgardner
December 12, 2011
| A version of this story appeared in
Volume 89, Issue 50

The common automobile tire contains rubber that’s extracted from latex-bearing trees and rubber that’s synthesized from petroleum feedstocks. Industrial biotechnology companies such as Genencor, Gevo, Amyris, and Genomatica want to give tire manufacturers a third option: biobased rubber ingredients made from sugar.

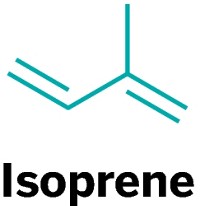
Microbial fermentation holds promise for making three renewable rubber intermediates: isoprene, isobutene, and butadiene. The compounds cover a wide swath of ground for the rubber-making industry. Five-carbon isoprene is used to make synthetic latex similar to that of the rubber tree. Isobutene and butadiene are four-carbon intermediates used to make butyl rubber and styrene-butadiene rubber.
Two leading tire makers—Goodyear and Michelin—along with synthetic rubber manufacturer Lanxess have entered into partnerships with industrial biotech firms to advance the commercial production of these rubber intermediates from sugar. They are motivated by tightening supplies of both natural and synthetic rubber, driven in recent years by strong global demand, especially from emerging economies.
New renewable sources will not be commercially available for another three to five years, but experts say the renewables won’t be too late to help alleviate what is becoming a supply headache for buyers. Today, the chemical intermediates come from the cracking of liquid feedstocks in ethylene plants. But as petrochemical makers switch to lighter natural gas feedstocks, production of C4 and C5 chemicals is drying up.
Even availability of natural latex rubber, which is used in large amounts in truck, aircraft, agricultural, and earthmover tires, has been limited because of soaring postrecession demand and constraints on the expansion of rubber plantation acreage. In 2010, natural rubber accounted for 34% of the value of raw materials purchased by Michelin, a substantial increase from 2009, according to the company.
The supply situation is unlikely to change, especially in North America, says William Hyde, director for C4 olefins and elastomers for the chemical consulting firm CMAI Global. “The expectation on the petrochemicals side is that we’ve only begun to see the impact of moving to lighter ethylene cracking. The rubber producer or tire maker steps back and says, ‘My supply situation is probably not going to strengthen much in the near term. What are my options?’ ”
What tire makers want is something to help them control volatile raw material costs. Prices of both synthetic and natural rubber jumped during the summer of 2011. The cost of a common grade of natural rubber shot up 55% in the second quarter compared with the year-ago quarter, while styrene-butadiene rubber prices increased 33%, according to the International Rubber Study Group, a trade organization that compiles statistics on rubber.
The group expects that overall global demand for both types of rubber will grow to 35.9 million metric tons by 2020, from 25.7 million metric tons in 2011. Demand will be met roughly equally by synthetic and natural rubber.
Hyde does not anticipate that microbes will steal a great deal of the market from petrochemical makers or rubber tree farmers. “I believe we will see commercial production,” he says. “To me the bigger question is not whether you are going to have commercially available material but whether it will be enough to make a material difference.”
David A. Benko, director of materials research and development at Goodyear, says the firm stands ready to use a great deal of sugar-derived isoprene in its tires. “Because it could replace natural rubber as well as synthetic rubber, that adds up to a lot of rubber that we can consume. We can get a good percentage of it into a tire.”
Launched in 2007, Goodyear’s partnership with biotech firm Genencor is the granddaddy of renewable rubber projects. Genencor genetically engineered microbes to produce the enzyme isoprene synthase, which is the enzyme that plants use to make latex from carbohydrates. In the summer of 2010, researchers from the firms reported that their microbes produced 60 g of isoprene per liter of sugar solution (Ind. Biotechnol., DOI: 10.1089/ind.2010.6.152).
“The way the collaboration works, Genencor works on the front end with its sugar feedstocks, fermentation, and genetic engineering,” Benko explains. “Goodyear brings its expertise with isoprene—we both make and use isoprene ourselves. So that makes up the whole package from beginning to end.”
To date, Goodyear has confirmed that biobased isoprene meets specifications for the catalysts it uses in rubber manufacturing and has even made concept tires with it. Originally, the project targeted 2013 for commercialization, but the time frame has been pushed back a few years. In May, DuPont acquired Genencor’s parent company, Danisco, and now the new owner is weighing in.
Prior to the acquisition, Danisco said it planned to invest $11 million in developing biobased isoprene in 2011. “The effort is strategically important to both companies, and we’re actively engaged in plans going forward—how best to do this, what scale, and when?” Benko says. Genencor executives declined to be interviewed for this story.
Fermentation-derived intermediates have reached a similar milestone at specialty chemical maker Lanxess and its partner Gevo. Biobased butyl rubber made from Gevo’s isobutyl alcohol has satisfied rigorous standards testing by the tire industry, according to the companies.
Gevo’s scientists engineered a yeast strain to produce isobutyl alcohol from sugar by blocking competing pathways for ethanol and acetic acid. Lanxess researchers added a dehydration process to convert isobutyl alcohol to isobutene, which can then be polymerized to butyl rubber. Lanxess has invested $27 million in Gevo and is now a major shareholder. The two firms are currently negotiating a supply agreement.
Christopher Ryan, Gevo’s president and chief operating officer, says the economics of the process are acceptable with corn sugar as a feedstock and look even better with sugar derived from cellulose. “When people look into the future, they may feel certain that oil prices will go up, but what happens to grain or sugar prices? You can’t answer that definitively.” With the promise of cellulosic feedstocks, he says, investors have more comfort that they are getting into a process with attractive economics in the long term.
In a newer partnership, French tire maker Michelin has joined with Amyris to develop renewable isoprene from sugar. Amyris says it will use technology similar to its process for making farnesene, a 15-carbon molecule that the firm plans to sell as an intermediate for lubricants and surfactants (C&EN, Dec. 5, page 26).
Meanwhile, renewable chemicals maker Genomatica told investors in August that it had made pound quantities of butadiene, a component of styrene-butadiene rubber, from renewable feedstocks. Genomatica is best known for making butanediol by feeding sugar to an engineered strain of Escherichia coli. The firm did not name any partners or potential buyers of its butadiene, but it did include plans for the product in its August filing for an initial public offering of stock.
Although isoprene has a head start over isobutene and butadiene, in all three cases the volumes produced in the next five to 10 years will remain quite small, CMAI’s Hyde cautions. In addition, he says, “it very much remains to be proven if they can produce on a cost-competitive basis [compared] with more traditional petrochemical pathways.”
But Goodyear’s Benko maintains that sugar-derived feedstocks will be valuable to cushion swings in raw material costs. “When we started the project, natural rubber was just hitting 70 cents per lb,” he says. “Now it’s up to $2.00. When you consume over 1 billion lb per year, that kind of pricing wreaks havoc.” Even worse, he points out, “a lot of markets are growing rapidly that require these raw materials. You can envision a time when you may not be able to get what you need.”



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter