Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Mass Spectrometry
Spying on early ovarian cancer with mass spec imaging
New technique tracks messenger molecules traveling between migrating cancer cells and ovarian tissue
by Louisa Dalton, special to C&EN
October 17, 2018

By the time ovarian cancer is discovered in a patient, the disease is often alarmingly advanced. That’s why less than half of women diagnosed with ovarian cancer survive past five years. And it’s why scientists would love to get a better understanding of how ovarian cancer starts and how it spreads. A new study uses mass spectrometry imaging (MSI) to do just that, by exposing the early chemical drivers of ovarian cancer (ACS Cent. Sci. 2018, DOI: 10.1021/acscentsci.8b00405).
A few years ago, researchers discovered that most ovarian cancers actually begin in a fallopian tube and make the ovary their second home. Joanna E. Burdette, a women’s health researcher at the University of Illinois, Chicago, wondered if some sort of early communication occurs between cancer cells in the fallopian tube and the ovarian cells. If so, Burdette didn’t have a technique for eavesdropping on that back-and-forth.
Fortunately, Laura M. Sanchez, Burdette’s colleague at UIC, specializes in tracking the small messenger molecules that shuttle between cells with MSI. Her snapshots reveal the position and mass-to-charge ratio of molecules in a three-dimensional tissue culture, creating “a molecular map of a biological sample,” she says.
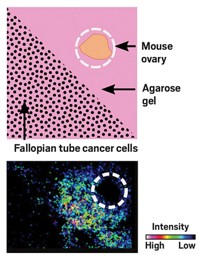
Using MSI, Sanchez, Burdette, and colleagues designed a stakeout of cancer cells’ communication with mouse ovaries. They removed ovaries from mice and placed them in wells of agarose gel with either cancerous mouse fallopian tube cells or normal fallopian tube cells. They let the cells grow for four days and then dried each sample flat for imaging with matrix-assisted laser desorption-ionization time-of-flight (MALDI-TOF) mass spectrometry.
Thirty-three signals corresponding to particular mass-to-charge ratios showed up significantly more frequently in wells holding cancer cells compared with wells of healthy cells. One particularly strong signal came from norepinephrine, a neurotransmitter released by neurons in the fight-or-flight response and also by the ovary as part of the ovulation cycle. Studies have linked stress-induced norepinephrine to aggressive ovarian cancer, yet researchers didn’t have a way of pinpointing norepinephrine’s source as the cancer takes hold.
To determine which cells were releasing norepinephrine—the fallopian tube cancer cells or the ovarian cells—the researchers divided a well and placed the ovary on one side and the cancerous fallopian tube cells on the other. After four days, they found norepinephrine fanning out from the ovary to the cancer cells.

The researchers suspect that the cancer cells are somehow hijacking the ovary cells to induce a come-hither message. That norepinephrine message could be triggered by an initial signal coming from the fallopian tube cancer cells during those first four days. They are now focusing on tracking that signal, as well as describing other signals in the mix.
Amanda B. Hummon of Ohio State University, who was not involved in the study, says the technique could be useful in her own research on colon cancer metastasis. “I think it is a beautiful example of developing an analytical method to go after a really important question in cancer biology.”
Advertisement
Sanchez believes the technique is an especially powerful tool for cancer research. Looking for chemical gradients in the tumor microenvironment “is a totally different way of looking at and thinking about cancer,” she says. Their setup can address any number of questions about cellular communication, she adds.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter