Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Separations
Natural protein captures rare-earth elements better than synthetic chelators
A lanthanide-binding protein found in bacteria could extract rare-earth metals more efficiently and with less waste
by Meredith Fore, special to C&EN
August 27, 2020

Rare-earth metals are essential parts of electronic components in phones, vehicles, displays, and wind turbines. A new study shows that a recently discovered protein from bacteria outperforms many current methods for extracting these elements from waste streams while potentially being more environmentally friendly (Inorg. Chem. 2020, DOI: 10.1021/acs.inorgchem.0c01303).
Concentrated amounts of rare-earth metals, also called lanthanides, are only found in a few ore deposits around the world. More abundant but far more dilute sources of these materials include electronic waste and rare-earth-containing coals. But these dilute sources are rarely economical to tap because the current methods for collection, which mainly involve solvent extraction, are inefficient and produce large amounts of toxic compounds.
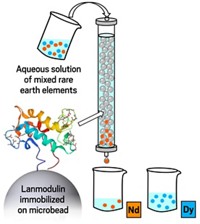
In 2018, Joseph A. Cotruvo Jr. of Pennsylvania State University and colleagues discovered a new protein, called lanmodulin, that selectively binds lanthanides in bacteria that require these rare earths to survive. In the new work, the team wanted to see if the protein could be useful in extracting these elements. They compared lanmodulin’s ability to pull lanthanides out of streams of electronic waste and rare-earth-containing coal with the traditional chelators used in current rare-earth processing plants. Lanmodulin was orders of magnitude more selective, capturing neodymium and europium ions while leaving behind ions present at higher concentrations in the waste streams, such as iron and zinc.
Lanmodulin “has a huge appetite for rare-earth metals and basically nothing else,” says study author Gauthier J.-P. Deblonde of Lawrence Livermore National Laboratory. “I’m not aware of any other macromolecules or small chelators which are able to display such universal selectivity for these elements.”
Most proteins denature at low pH or high temperature, but lanmodulin proved to be unusually robust. Rare-earth ions bound to lanmodulin at a pH as low as 2.5 and temperatures up to 95 °C, which Deblonde calls “completely unique” to this protein. Synthetic protein chelators typically release their lanthanides at about pH 6, which can limit their industrial applications.
The advantage of the lanmodulin protein’s powerful lanthanide selectivity is that it works in an aqueous environment, a greener option than typical solvent extraction procedures that produce difficult-to-dispose-of contaminated organic waste.
“Compared to the synthetic proteins that have been tried for lanthanide binding, this protein just blows them out of the water,” Cotruvo says. “And the organisms it comes from grow on plants, in soil—they’re ubiquitous.”
“It’s just a breathtaking development for this totally new field of rare-earths biochemistry,” says Eric J. Schelter of the University of Pennsylvania, who was not involved in the study. “I think it has potential to contribute to the goal of making rare-earth mining more sustainable because it’s just so incredibly selective.”




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter