Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Diiron oxygenase cleaves C-H bond in an unexpected way
May 1, 2006
| A version of this story appeared in
Volume 84, Issue 18
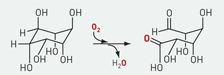
An iron-containing oxygenase has been found to use an unconventional strategy for cleaving C−H bonds. J. Martin Bollinger Jr., Carsten Krebs, and coworkers at Pennsylvania State University report that the enzyme myo-inositol oxygenase (MIOX) operates by a mechanism different from that of related diiron enzymes. The pair of iron atoms in MIOX uses O2 to cleave a single C−H bond in the cellular messenger molecule myo-inositol to produce d-glucuronate (shown). The researchers find that MIOX starts its catalytic cycle with one iron as Fe(II) and the other as Fe(III), unlike all of its diiron oxygenase brethren, which start with both irons in the +2 oxidation state (Biochemistry 2006, 45, 5393 and 5402). This suggests that MIOX's Fe(II)/Fe(III) cofactor is unlikely to go on to generate the high-valent Fe(IV)=O species its relatives use to oxidize their substrates, Bollinger says. The researchers proved their hunch by using deuterium-labeled substrate and electron paramagnetic resonance spectroscopy, showing that the oxygenating species in MIOX is in fact an Fe(III)/Fe(III) superoxide species (Proc. Natl. Acad. Sci. USA 2006, 103, 6130).


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter