Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Materials
Artisan Molecules
Metal-terpyridine complexes form fractal polymers with aesthetic shapes
by Stephen K. Ritter
May 15, 2006
| A version of this story appeared in
Volume 84, Issue 20

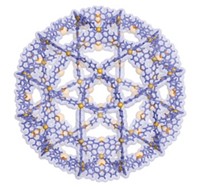
Molecular self-assembly has allowed chemists to become ever more creative by incorporating aesthetically pleasing patterns into the structures of macromolecules. In one of the latest examples, a team of chemists led by George R. Newkome at the University of Akron, in Ohio, has prepared a "Sierpiski hexagonal gasket," the first example of a molecule whose structure has a nondendrimer fractal pattern (Science, published online May 11, dx.doi.org/10.1126/science.1125894).
Fractals are mathematical constructs used to describe "self-similar" structural motifs, which appear similar at all scales of magnification. Some examples are snowflakes and tree branches, the latter pattern already well-known in chemistry from polyamidoamine and other dendrimers. The Sierpiski gasket, named after Polish mathematician Wacaw Sierpiski (1882-1969), is a hexagonal array of hexagons. Sierpinski derived an equation that describes how the shape can be formed from a series of interrelated equilateral triangles.
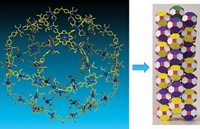
Newkome and his colleagues mimicked the gasket pattern by inducing sets of ruthenium and iron bis(terpyridine) complexes to self-assemble. They first built a hexagonal ring of six ruthenium complexes joined together by benzene rings. This hexamer has exposed terpyridine groups on two corners, and its reaction with FeCl2 forms iron bis(terpyridine) complexes that lash the hexamers together into a larger hexagon.
Altogether, the synthesized gasket has 36 ruthenium complexes and six iron complexes. The gasket is large enough for the individual hexamers to be visible in scanning tunneling microscope images. Beyond the artistic value, fractal polymers have potential uses as magnetic and electronic materials.
The synthesis "moves dendrimers from being the unique member in the chemical fractal family to that of a subset of fractal materials," Newkome says. "In essence, we now have dendritic and nondendritic fractal polymers."



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter