Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Physical Chemistry
Mn4Ca cluster shapes up
November 6, 2006
| A version of this story appeared in
Volume 84, Issue 45
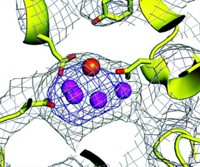
A new window on the structure of the metal cluster that splits water into O2, H+, and electrons during photosynthesis should help those aiming to mimic nature's ability to harvest energy from the sun (Science 2006, 314, 821). Previous structural and spectroscopic studies have failed to nail down the structure of photosystem II's Mn4Ca cluster. Johannes Messinger of Max Planck Institute for Bioinorganic Chemistry in Mülheim, Germany; Athina Zouni of the Technical University Berlin; and Vittal K. Yachandra of Lawrence Berkeley National Laboratory now provide a more satisfying snapshot of the cluster (shown in its protein environment; red balls = Mn, gray balls = bridging O, green ball = Ca). They obtained the structure by first using the protein's X-ray diffraction pattern to align a single crystal in an X-ray beam and then using X-ray absorption fine-structure spectroscopy to measure Mn-Mn and Mn-Ca vectors. The team plans to use the same method to probe subtle changes in the cluster during catalysis.






Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter