Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
The Riches Of Gold Catalysis
Chemists are using this precious metal to catalyze a treasure trove of reactions
by Raychelle Burks
September 24, 2007
| A version of this story appeared in
Volume 85, Issue 39
GOLD, ONCE CONSIDERED catalytically dead, is dazzling chemists with the repertoire of reactions it can catalyze. From simple oxidations to complex organic synthesis, gold has proven to be a powerful and multipurpose catalyst. Gold catalysts already have made it out of the lab and into commercial use in the air quality and alternative energy industries, and they are poised to make a splash in the fine chemicals market next.
Gold catalysts come in two types: heterogeneous and homogeneous. Heterogeneous gold catalysts exist in a different phase from the reactants and products and typically consist of tiny particles of gold or a gold alloy attached to a solid support. Homogeneous gold catalysts, on the other hand, are normally monometallic complexes with organic ligands and exist in the same phase as the reactants and products.
Because they can catalyze valuable reactions such as carbon-carbon bond formation—and because their reactivity can be tuned simply by tweaking the organic ligands that surround the metal—homogeneous gold catalysts have become increasingly powerful tools for organic synthesis. But their commercialization has lagged behind that of heterogeneous catalysts, which are already used commercially to oxidize a variety of substrates, including carbon monoxide found in specialty gases and car exhaust.
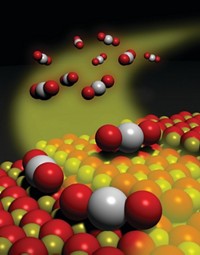
Chemist Masatake Haruta, now at Tokyo Metropolitan University, was the first to use gold to remove trace CO by oxidizing it to CO2 (Chem. Lett. 1987, 16, 405). Such gold-catalyzed CO oxidation "really caught people's imagination because it's a very easy reaction for everybody to do," says Graham Hutchings, chemistry professor at Cardiff University, in Wales, who specializes in gold catalysis.

Haruta's catalysts of ultrafine gold particles supported on metal oxides proved more active and stable than the supported manganese or copper catalysts that were then routinely used for CO oxidation. "Haruta was the one who really discovered nanoparticle gold catalysis," says D. Wayne Goodman, codirector of Texas A&M University's Center for Catalysis & Surface Chemistry. Haruta's work sparked academic research and industrial applications into supported nanosized gold particle catalysts for CO oxidation.
Such catalysts have found their way into CO-removing safety masks designed for firefighters. The masks rely on nanosized gold-supported catalysts to oxidize and thus minimize the user's exposure to this poisonous gas. Union Chemical Laboratories and Novax Material & Technology, both of Taiwan, currently market such masks, and 3M plans to begin selling a similar product soon.
Gold-based oxidation catalysts are also finding use in the purification of hydrogen gas for use in fuel cells. Normally, the process by which H2 is produced from hydrocarbon fuels leaves residual amounts of CO in H2 gas streams. Because a hydrogen fuel cell's anode can be rendered useless by CO, removing CO from the H2 is essential.
Oxidative removal of CO presents a challenge, Goodman explains, as standard platinum catalysts will oxidize both the CO and H2 indiscriminately. For selectivity, researchers have turned to gold. In experiments designed to model a fuel cell as it might be used in an electric vehicle, Hutchings' research team demonstrated that Au/Fe2O3 catalytic systems can be used to selectively oxidize 99% of the CO in the presence of H2 (Chem. Commun. 2005, 3385). Catalyst manufacturer Haldor Tops??e and others have applied for patents on gold-based oxidation catalysts for H2 purification.
Catalytic converter manufacturers are turning to gold-based oxidation catalysts to combat diesel fuel emissions. Earlier this year, catalyst maker Nanostellar of Redwood City, Calif., introduced an Au-Pt-Pd oxidation catalyst for use in light- and heavy-duty diesel engines. Like pure platinum catalysts marketed for cleaning up diesel emissions, Nanostellar's gold alloy scrubs CO and hydrocarbons from diesel emissions via oxidation. With gold currently selling for roughly half the cost of platinum, Pankaj Dhingra, Nanostellar's chief executive officer, says that his company's gold alloy catalyst will "improve the cost equation for the diesel industry," which currently is faced with federal regulatory pressures to clean up emissions.

GOLD CATALYSTS' oxidative powers soon may also find use in the fight against mercury emissions from coal-fired power plants. Last year, the U.S. Department of Energy awarded San Francisco-based engineering firm URS $2.5 million to explore whether the firm's supported gold catalyst could be used to remove mercury from coal-fired power plant emissions. The gold catalyst oxidizes any Hg(O) present; the oxidized mercury is then captured and prevented from escaping from the plant.
Researchers are also eager to harness the selectivity of heterogeneous gold catalysts to make commercially important catalytic processes more environmentally friendly. Haruta points to alcohol oxidation as a key example of an important industrial reaction that gold is poised to turn green.
Haruta's enthusiasm stems from recent reports of gold-catalyzed alcohol oxidation from two academic groups. Avelino Corma, of Polytechnic University in Valencia, Spain, and coworkers used a solid-supported Au-CeO2 catalyst to selectively oxidize alcohols to aldehydes and ketones under O2 at atmospheric pressure (Angew. Chem. Int. Ed. 2005, 44, 4066). This reaction requires neither solvent nor base, and yields are equal to or better than what is achieved with commonly used palladium-supported catalysts. And last year, Hutchings showed that a Au-Pd solid-supported catalyst can ramp up alcohol oxidation turnover 25 times over previously reported values, giving the desired aldehyde product in 90% yield (Science 2006, 311, 362).
One company that has begun using heterogeneous gold-catalyzed alcohol oxidation commercially is Japanese chemical manufacturer Nippon Shokubai. The company built a pilot plant that can produce, on a monthly basis, tons of methyl glycolate, which, among other uses, serves as a solvent in some semiconductor manufacturing processes, a building block for cosmetics, and a cleaning agent for boilers and metallic surfaces. The process uses a proprietary supported Au-Pb catalyst to oxidize ethylene glycol to methyl glycolate in methanol (Catal. Today 2006, 117, 210).
Although heterogeneous gold catalysis has attracted industrial attention, full-scale industrial use has yet to emerge. "These things always take longer than you might imagine," says Richard Holliday, head of industrial applications at the World Gold Council, a trade association of the leading gold mining companies.
There may be more potential industrial applications of such catalysts in the future. Researchers have begun to explore the utility of heterogeneous gold catalysis in reactions beyond simple oxidations. For example, Corma and colleagues used supported gold catalysts in the cycloaddition of o-alkynylbenzaldehydes with alkynes (Top. Catal. 2007, 44, 237). The heterogeneous gold catalysts pulled off this carbon-carbon bond-forming reaction in 99% yield.
Aside from this recent example, however, homogeneous gold catalytic systems dominate over heterogeneous ones in organic synthesis.
Yoshihiko Ito and colleagues at Kyoto University, in Japan, pioneered the use of gold homogeneous catalysts in organic synthesis two decades ago by showing that a chiral ferrocenylphosphine-Au(I) catalyst can catalyze asymmetric aldol reactions (J. Am. Chem. Soc. 1986, 108, 6405). But homogeneous gold catalysis didn't take off in organic synthesis until J. Henrique Teles and colleagues at BASF, in Germany, demonstrated that the reactivity of such gold catalysts could be fine-tuned by altering ligands (Angew. Chem. Int. Ed. 1998, 37, 1415).
Teles' team synthesized various L–Au(I) catalysts (where L = phosphane, phosphite, or arsine) and studied their ability to catalyze the addition of alcohols to acetylenes. The researchers found that changing L influenced both catalytic activity and product yield. "It turned out that only very low catalyst loadings were necessary and very fast reaction rates were possible" with the L–Au(I) catalysts, Teles says. Though the process never went commercial, the ability to fine-tune gold's catalytic power through ligand selection further burnished gold homogeneous catalysts' reputation in organic synthesis.
MORE RECENTLY, F. Dean Toste, a chemistry professor at the University of California, Berkeley, has furthered efforts to tune the metal center of homogeneous gold catalysts by ligand selection. Toste has been exploring Au(I) bearing phosphine (R3P) ligands. For a variety of intra- and intermolecular organic reactions, Toste has shown that product yields vary with R (J. Am. Chem. Soc. 2004, 126, 4526 and 15978). The phosphine can carry a single type of R group or a complex mixture, Toste notes. Not only can the reactivity of these R3PAu(I) catalysts be tuned, but the catalysts are also robust and require mild reaction conditions.
Au(III) catalysts such as AuCl3 also have proven successful at catalyzing a variety of organic reactions. AuCl3 captured chemists' attention in 2000 when A. Stephen Hashmi, now at the University of Heidelberg, in Germany, showed that the homogeneous AuCl3 catalysts could catalyze both intramolecular cyclizations and cross-cycloisomerization reactions (Angew. Chem. Int. Ed. 2000, 39, 2285 and J. Am. Chem. Soc. 2000, 122, 11553).
For gold-catalyzed organic synthesis, simple and substituted alkynes are popular substrates. "Phosphine gold(I) complexes have been shown to be the most reactive catalysts for the activation of alkynes," says Antonio M. Echavarren of the Institute of Chemical Research of Catalonia, in Spain. Activation of alkynes allows for the "development of new reactions for the ready buildup of molecular complexity," Echavarren explains. He used a bulky R3PAu(I) catalyst for cycloadditions of enynes with alkenes to build complex bi- and tricyclic compounds (J. Am. Chem. Soc. 2005, 127, 6178).
Gold is gaining ground in organic synthesis, but there are still areas to conquer. "Despite some progress, asymmetric synthesis with chiral gold catalysis is relatively underdeveloped," Echavarren says. Development of such catalytic systems would open a new door "to the synthesis of natural products and pharmacologically active compounds," says Norbert Krause, a chemistry professor at the University of Dortmund, in Germany. He recently used Au(I) and Au(III) catalysts for stereoselective cycloisomerization of various β-hydroxyallenes to the corresponding chiral dihydropyrans, a common structural unit in natural products and other biologically active compounds (Org. Lett. 2006, 8, 4485).
Toste's team recently introduced a unique approach to gold-catalyzed enantioselective reactions. His group demonstrated that by using chiral phosphate counterions in concert with phosphine-Au(I) catalysts, they could achieve enantiomeric excess values of 90 to 99% in the cyclization of substituted allenol substrates (Science 2007, 317, 496; C&EN, July 30, page 17). For a particularly difficult asymmetric transformation, the team combined a chiral counterion and chiral phosphine-Au(I) catalyst. With an enantiomeric excess of 82%, this reaction illustrates the synergistic effect of combining chiral counterions with chiral metal catalysts, Toste notes.
These achievements in asymmetric catalysis could help usher homogeneous gold catalysts into industrial settings. "To the best of my knowledge, there are no industrial applications of homogeneous gold catalysis at present," Krause says. He thinks asymmetric gold catalysis will tip the scales to commercial use in the fine chemicals and pharmaceutical industries.
Gold catalysts are already commercially available from an industry giant. Sigma-Aldrich offers an array of gold precatalysts and has a website dedicated to gold catalysis. "We believe that gold catalysis will play a key role in chemical synthesis in the coming years as evidenced by the growing number of publications in this emerging field," says John Chan, market segment manager of chemical synthesis at Sigma-Aldrich.
Now that gold catalysts are firmly entrenched in organic synthesis and oxidation chemistries, interest in developing theory to explain their reactivity is growing. A better theoretical understanding of reactivity is expected to aid the design of improved catalysts. But because heterogeneous and homogeneous gold catalysts differ in structure, each catalytic system requires its own theoretical treatment.
IN HETEROGENEOUS catalysis, gold is considered the best example of "nano effects," the observation that a material's properties can change drastically when particle sizes get down to the nano regime. "Gold is inert macroscopically," Goodman notes, "yet when you make it nanoscale, it becomes extremely interesting" and special in terms of electronic properties. At particle diameters less than 2 nm, which corresponds to particles containing fewer than 300 atoms, Haruta notes, "there is a dramatic change in electronic structure owing to the quantum size effect."
Advertisement
Toste points to quantum mechanics and even special relativity to explain the reactivity of homogeneous phosphine-Au(I) catalysts (Nature 2007, 446, 395). His research indicates that relativistic effects on some of the faster moving electrons in the 5s and 6p orbitals could be causing the s orbital to contract, thereby shielding the p orbital so that it can expand.
Such changes, he and his colleagues theorize, could render Au(I) catalysts strong Lewis acids and explain their ability to stabilize cationic reaction intermediates. Echavarren concurs, adding that his work shows that relativistic effects underlie the superior π-acidity of Au(I) complexes that makes them superior catalysts for alkyne activation.
Hashmi, whose lab is working to learn more about the reactivity of Au(I) and Au(III) complexes in homogeneous catalysis, notes that despite recent progress in this area, further work must be done to more fully understand gold's reactivity.
With academic and industrial researchers alike hotly pursuing new catalysts and applications, gold has proven to be a powerful catalyst with surprising versatility. "I think it's taken everybody by surprise. It makes people think 'How could I have missed it for all these years?' " Hutchings says. No longer overlooked, gold's future in catalysis seems bright.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter