Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Materials
In Search of Concrete Evidence
Materials scientist suggests parts of the Egyptian pyramids were made from reconstituted limestone
by Bethany Halford
February 11, 2008
| A version of this story appeared in
Volume 86, Issue 6

EVERY DAY on the outskirts of Cairo, thousands of tourists brave the dust and heat of the Egyptian desert to gaze upon the Great Pyramids of Giza. To say the pyramids are colossal does not even begin to describe the sheer magnitude of these man-made mountains. The largest—the Great Pyramid, built by the pharaoh Khufu—is nearly 50 stories high and sits on a base the size of 10 U.S. football fields. But the numbers simply don't do justice to these giants or to the enormous feat of engineering it took to create them more than 4,500 years ago.
Michel W. Barsoum, a materials science professor at Drexel University, grew up in the shadow of the ancient pyramids. To Barsoum, the pyramids have always been a symbol of his native Egypt, a wondrous example of his ancestors' engineering acumen. He never imagined that one day he would be using high-tech instrumentation and his knowledge of materials science and chemistry to challenge the theories that generations of Egyptologists have held about how the pyramids were built.
"This is not my day job," Barsoum says. It's the first thing he wants people to know before they hear about his pyramid research. Passions run high when it comes to the pyramids, and he is at pains to make it clear that the focus of his research—high-tech carbides and nitrides—has absolutely nothing to do with them. "I have no vested interest in this," he says. "I think it's a really neat problem, and that's why I'm working on it."
Barsoum's saga began six years ago, when he got a call from a retired engineer and pyramid enthusiast named Mike Carrell who asked if he was familiar with the mystery of the pyramids. Did Barsoum know that the Great Pyramid contains some 2.3 million blocks, averaging 2.5 tons each? Did he know that Egyptologists estimate that it took 23 years to construct that pyramid? Did he realize that, according to a back-of-the-envelope calculation, one massive block would need to have been laid into place every six minutes, assuming the builders worked around the clock for more than two decades? Did he know that some blocks in the Giza pyramids are so close together that it's impossible to slip even a piece of paper between them?
Then Carrell told Barsoum that the great mystery of how the pyramids had been constructed had already been solved. He said the French geochemist Joseph Davidovits had postulated decades ago that the pyramids had not, as Egyptologists thought, been made of limestone carved by copper tools into great blocks that were then hoisted into place by levers. Rather, Davidovits argued, they had been cast from reconstituted limestone concrete.
The explanation, Barsoum says, made him laugh out loud. If this theory held any weight, why had no one gathered any scientific evidence to prove it? He said to Carrell, "If I can't tell you the difference between natural limestone and artificial limestone in about two hours on a scanning electron microscope, then shame on me."
IN HINDSIGHT, Barsoum recognizes his hubris in taking on the project. "What was supposed to take me two hours on the scanning electron microscope turned into a five-year odyssey," he says. To complete their analysis, Barsoum and his colleagues had to take more than 1,000 micrographs, focusing their scanning electron and transmission electron microscopes on samples taken from the Great Pyramid and samples of natural rock from which the pyramids are thought to have been constructed.
According to Davidovits' theory, the ancient Egyptians used natural limestone rubble combined with clay in a high-pH solution to create their concrete. "Because all clays contain sodium and aluminum, we started looking for sodium and aluminum in the cementing phase of the pyramid samples," Barsoum explains. "We didn't find any sodium, and we didn't find any aluminum. The only thing we found was silica."
It's common to find silica in limestone, so Barsoum's initial findings would seem to indicate that Davidovits' theory, or at least his recipe, was incorrect. But Barsoum also found chemical evidence that the natural stone differed significantly from stone taken from the pyramids.
In all the samples taken from the pyramids he found sulfur, but he found no trace of the element in the natural stone samples. The stone taken from the pyramids appears to be significantly more hydrated than natural limestone from the region.
Barsoum also found that portions of the pyramid samples appeared to be amorphous; their structure is highly disorganized. It would be very unusual, he points out, to find such disorganization in natural sedimentary stone formed on a geological timescale. Furthermore, the silica in one of the pyramid samples was in the form of nanospheres—another structure that he says doesn't form naturally in limestone.
In the same pyramid sample, Barsoum notes the presence of a layer of calcium phosphate, or bone. The Egyptians had to mix calcium phosphate with some type of geological binder to make it solidify and stick to a limestone block. He and his colleagues note the presence of this binder not just in the calcium phosphate layer but also deep within the limestone beneath the calcium phosphate. "I cannot come to you with more convincing evidence of casting. After that, I'd need to get you a videotape of them doing it," Barsoum says, referring to the ancient pyramid makers.
That's not to say the whole of the pyramids were cast, Barsoum emphasizes. He believes, on the basis of his research, that only portions of the pyramids—their tops, backing blocks, and outer and inner casings—were made from reconstituted limestone. The pyramids' cores, he thinks, were most likely made from cut limestone.
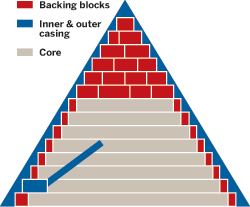
Barsoum believes the pyramids' tops, backing blocks, and inner and outer casings were cast from reconstituted limestone. Their cores, he says, are probably composed of carved limestone blocks.
"I think Barsoum has certainly made the case that there is something in some of the stones that is not consistent with them being just straight limestone," remarks David Walker, a geology professor at Columbia University. "I think it's also clear that geologists looking at least at some of those rocks would have no question, no hesitation, not even a glimpse of a doubt that they really are actually cut limestone. But that doesn't mean they all are," he says.
It took Barsoum more than two years to publish his results (J. Am. Ceram. Soc. 2006, 89, 3788). His report grabbed headlines and quickly prompted a backlash from both the geology and Egyptology communities. Barsoum is still having trouble publishing a follow-up paper. It has been, he says, a lesson in "political science."
"THE IDEA of concrete pyramids is preposterous," says Robert L. Folk, a professor of geology at the University of Texas, Austin, who has studied the pyramid stones. "It is well-known that the Egyptians applied some sort of coating to surfaces they wanted to paint on. I suspect Barsoum has been looking at this outer coating. He needs to get a few centimeters deep into the stone."
Folk also discounts any conclusions drawn from the presence of silica nanospheres in the sample. Silica nanospheres, he says, occur in opal, a geological constituent of the pyramid stones.
Critics also point to Barsoum's small sample size. He agrees that the small number of samples he studied likely skews his results, but notes that he had difficulties procuring official pyramid samples from Egypt's Supreme Council of Antiquities.
"There is a natural mistrust of unusual theories in the Egyptology community," Barsoum says. There would have to be, he adds, considering some of the strange ideas people have about the pyramids—such as that they were nuclear power plants or giant batteries. Hear enough of these oddball theories, Barsoum says, and you're bound to be suspicious of anyone outside of the establishment.
Still, Barsoum believes such suspicion is unfounded in the case of his findings. If parts of the pyramids were made from an ancient concrete, he argues, it doesn't diminish the achievement of their construction. Rather, he says, it means the ancient Egyptians discovered concrete 2,000 years before the Romans. "That a lime-based cement cast and cured at room temperature would survive for 4,500 years, whereas the best our civilization has to offer—Portland cement—under the best of circumstances lasts 150 years or less, is both awe-inspiring and humbling," Barsoum concludes.





Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter