Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Extreme C–C Bonds
Highly strained cyclopropane-based molecules help chemists refine ideas about chemical bonding
by Stephen K. Ritter
May 11, 2009
| A version of this story appeared in
Volume 87, Issue 19
stabilize the strained tetrahedrane framework via the central C–C bond (red).

stabilize the strained tetrahedrane framework via the central C–C bond (red).
THE CARBON-CARBON BOND is arguably the most fundamental connection in all of chemistry. Yet a century after chemists began in earnest to unravel the mysteries of chemical bonding, the intricate details of C–C bonds are still unfolding.
Three research teams have recently contributed to the growing understanding—and growing complexity—of C–C bonding with reports on unique cyclopropane-based compounds.
In one case, Akira Sekiguchi and coworkers at the University of Tsukuba, in Japan, reported the synthesis of the first examples of aryl-substituted tetrahedranes (J. Am. Chem. Soc. 2009, 131, 3172). These compounds exhibit unusual electronic properties made possible by a stabilizing C–C bond that joins the geometrically strained tetrahedrane and the aryl substituent.
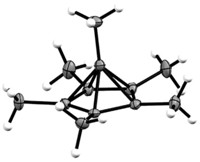
Tetrahedranes are pyramid-shaped C4 compounds that in essence consist of four fused cyclopropane rings. Chemists consider the parent tetrahedrane, C4H4, to be one of the most highly strained organic molecules yet conceived. The compound is so exceedingly unstable that chemists haven't been able to make it. Sekiguchi's group and the group of Günther Maier of Justus Liebig University, in Giessen, Germany, have shown that tetrahedranes supported by bulky substituents such as tert-butyl or trimethylsilyl are stable.
"Tetrahedrane is very unstable due to its high cage strain and high-energy C–C bond orbitals," notes computational chemist Yirong Mo of Western Michigan University, who has studied the bonding in tetrahedrane and its derivatives. Although tetrahedrane technically is a saturated molecule, it acts like it's unsaturated, possibly because of "three-dimensional aromaticity" of the cagelike σ-bond network, Mo says.
One way to stabilize the compound is by forming a bond between tetrahedrane and a large substituent, which provides a conduit for electrons to flow and offers extra stability through a σ-π hyperconjugation effect. Mo has projected that adding substituents with better electron-donating abilities to tetrahedranes should enhance hyperconjugation and thus stability, which could be observed in the form of shorter C–C bond lengths.
Sekiguchi and coworkers took a crack at doing that by adding unsaturated groups to trimethylsilyl-substituted tetrahedrane. Their initial attempts with phenyl groups failed because the electron-donating properties don't appear to be sufficient. But when the researchers used fluorinated phenyl groups, they successfully made aryl-substituted tetrahedranes.
The C–C bond joining a single perfluorophenyl group with the trimethylsilyl-substituted tetrahedrane is 1.450 Å, shorter than the usual 1.507-Å bond observed in other examples of sp3-sp2 hybridized bond systems, Sekiguchi and coworkers note. When the perfluorophenyl group has an ethynylphenyl substituent, which supplies additional electron density into the tetrahedrane-aryl system, the bond is shorter at 1.438 Å, supporting Mo's prediction.
In a second case of unique C–C bonding, Guy Bertrand and coworkers at the University of California, Riverside, devised a plan to synthesize a stable triafulvalene—the smallest and most strained member of the fulvalene family of compounds (Angew. Chem. Int. Ed. 2009, 48, 517).
Fulvalenes are a broad class of hydrocarbons consisting of two unsaturated rings joined through a C=C bond. Although many examples of fulvalenes with different ring sizes are now known, the simplest fulvalene, composed of two cyclopropene rings, has never been made. Like the parent tetrahedrane, it is too unstable to stand on its own.
Bertrand and coworkers turned to bulky aromatic ring substituents to lend a helping hand. At first, they tried to add two triisopropylphenyl groups to a chlorinated cyclopropene, with the idea of coupling two of the cyclopropene rings to make the triafulvalene. But two triisopropylphenyl groups are too big to fit around cyclopropene.
The researchers resorted to using just one triisopropylphenyl along with a trimethylphenyl (mesityl) group. With their bulkily substituted dichlorocyclopropene in hand, Bertrand and coworkers reduced it with magnesium metal, which coupled two of the molecules to form the substituted triafulvalene derivative they were after.
The crystal structure of the compound reveals that the C=C bond joining the two cyclopropene rings is exceedingly short, even for a double bond, at only 1.303 Å. The bond could be shorter still perhaps, Bertrand's team notes, but the bond angles of the bulky substituents suggest that conjugation between the aromatic rings and the fulvalene system is weak. In addition, other C–C bond lengths in the cyclopropene rings indicate that the π bonding is highly localized around the C=C bonds.
It's not surprising then that the substituted triafulvalene is quite reactive, Bertrand says. At room temperature, water adds across the central C=C bond to form the HC–COH adduct. "This is certainly one of the very rare examples of spontaneous addition of water to the C=C bond of a hydrocarbon," Bertrand points out.
In a third case of unusual C–C bonding, a multinational team of computational chemists has taken a fresh look at the bonding in [1.1.1]propellane, a propeller-shaped C5 molecule made up of three fused cyclopropane rings. Chemists have been intrigued by the nature of the central bridging C–C bond in propellanes, in some cases challenging the notion that the bond exists at all. Now, Sason Shaik of Hebrew University of Jerusalem, in Israel; Philippe C. Hiberty of the University of Paris, South; Wei Wu of Xiamen University, in China; and coworkers think they have a plausible explanation for the bonding based on ab initio valence bond calculations (Angew. Chem. Int. Ed. 2009, 48, 1407).

The researchers became interested in propellanes as possible examples of molecules exhibiting "charge-shift bonding," a potentially new paradigm in bonding. Charge-shift bonding is not the result of traditional sharing of an electron pair between two atoms in a covalent bond or the transfer of an electron from one atom to another in ionic bonding, Shaik explains. In fact, charge-shift bonds are characterized in part by depleted electron density between atoms. The bonding interaction instead arises primarily from the stability gained by resonance between covalent and ionic components, he says.
CHARGE-SHIFT BONDING is a controversial topic among theorists, so the only way for Shaik and his colleagues to convince the chemistry community of its existence is to find real-world examples. So far they've found charge-shift bonding only in compounds with highly electronegative elements that are rich in electron lone pairs (like F2)—and now in highly strained molecules like propellanes. In [1.1.1]propellane, the bonding electrons of the central C–C bond experience repulsion from the peripheral C–C bonds. The central C–C bond exhibits all the properties of a charge-shift bond, whereas the remaining C–C bonds are normal covalent bonds, Shaik says.
One could imagine that unusual charge-shift bonds could contribute to the structures of other highly strained polycyclic compounds, such as tetrahedranes and fulvalenes, Sekiguchi observes. In fact, after learning about Sekiguchi's tetrahedrane work and Bertrand's triafulvalene, Wu, Shaik, and Hiberty ran calculations on the C–C bonds in those compounds.
In both cases, the bonds turn out to be traditional covalent bonds because they are not subject to repulsion like [1.1.1]propellane, Shaik says. In short, cyclopropane's 60° bond angles require the C–C σ bonds to have high s-orbital character, which is observed in the shorter than normal bond lengths. The π bonds are impacted as well, Shaik adds. As bond lengths get shorter, the π-bond covalent-ionic resonance increases and edges toward becoming a charge-shift bond, but doesn't quite get there, Shaik says.
Beyond better understanding the C–C bonds, all these examples of cyclopropane-based compounds have peculiar spectroscopic features and unusual reactivity, Bertrand concludes. "Taming these molecules might one day lead to materials with novel physical and chemical properties."





Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter