Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Analytical Chemistry
Keepers Of The Gate
Advanced imaging and detection technologies help root out threats of terrorism in airports
by Mitch Jacoby
June 1, 2009
| A version of this story appeared in
Volume 87, Issue 22

WITH ALMOST 2 million travelers passing through U.S. airports daily, securing air travel against the threat of terrorist explosives is a colossal job. Sometimes security-checkpoint lines at busy airports creep along at a snail's pace. But by and large, a huge number of passengers and a much larger number of bags and parcels move through the security screening process rather quickly.
At the heart of that process lie analytical tools based on X-ray computed tomography, ion-mobility spectrometry, and other imaging and trace chemical detection technologies. These tools enable officers of the Transportation Security Administration (TSA) to quickly screen passengers and their bags for concealed weapons and bombs, as well as trace quantities of explosives and other illicit substances. Researchers and instrument manufacturers are working to boost the analytical power of these workhorse technologies and to develop new detection methods that will further enhance air-travel security while making the screening process more expedient.
Screening passengers and their bags for explosives is "an enormously challenging detection problem," TSA Chief Scientific Adviser Lyle O. Malotky says. The task is akin to searching for a needle in a haystack but worse because of the enormous variety of things people bring aboard airplanes. Clearing business travelers through security is often straightforward. Those frequent flyers might come to an airport with little more than a few shirts and socks and a couple of personal items in a commuter bag. But other passengers come with suitcases stuffed with electronic devices, small appliances, and even automobile parts, Malotky says.
Screeners must differentiate among all those items to determine whether passengers are concealing explosives or hazardous materials. And TSA officers need to do so quickly and thoroughly. Underscoring the need for thoroughness, Malotky says that even now with the public so well-informed about air-safety procedures, TSA still finds about 20 firearms per week at airport checkpoints.
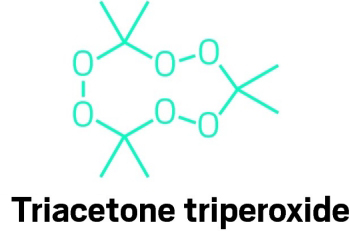
The security agency's task is further complicated by the large number of explosive materials that can be used to make bombs. A cursory check of the Internet reveals a huge number of articles and videos guiding users through kitchen-chemistry procedures for synthesizing explosives and fashioning bombs from products commonly available at hardware and drug stores. Dozens of amateur videos, for example, demonstrate the latent power of triacetone triperoxide (TATP or acetone peroxide), an explosive tied to Richard Reid, the "Shoe Bomber," and the July 2005 London bombings. That compound is readily made from acetone, hydrogen peroxide, and mineral acid. "How-to" guides on the Internet also teach users to make various organic peroxides, nitroamines, and other potent explosives at home.

"THE GROWING ABILITY to make improvised explosive devices out of fairly common chemicals means that screeners need to detect a wider spectrum of materials than ever before," says Susan F. Hallowell, director of the Transportation Security Laboratory, in Atlantic City, N.J. The lab develops and validates methods and tools for detecting explosives and other types of threats for the Department of Homeland Security. Hallowell explains that the challenge is to develop instruments and procedures for detecting an expanding portfolio of analytes with high sensitivity in a way that provides an operator with the information to make a security decision quickly but with a low false-alarm rate.
X-ray computed tomography (CT) serves as a cornerstone of security screening. Large airports, such as Chicago's O'Hare, make use of "in-line" CT instruments to scan thousands of checked bags per hour for explosives. By irradiating luggage with intense X-ray beams and measuring the extent of beam attenuation, these instruments probe the density and other properties of the bags' contents. Then, on the basis of automated algorithms that evaluate those data and compare them with values for explosives, the machines forward the bags for loading onto aircraft or hold them for further inspection.
About 85% of bags pass security automatically, Malotky says, because the densities and other properties of explosives tend to be different from most items typically found in a suitcase. For the other roughly 15% of checked bags, TSA officers intervene and examine the images manually, sometimes adjusting contrast or altering the angle to get a clearer view of the suspicious contents. If the officer remains concerned about a bag, the officer will open, inspect, and screen it for both visible and trace quantities of explosives (using swab tests), and depending on the outcome, will send it off to be loaded onto an airplane.
To boost throughput and reduce the false-alarm rate, U.K.-based Smiths Detection and other manufacturers design their latest baggage scanners to probe luggage with multiple X-ray beams and at more than one X-ray energy. Mark Laustra, a vice president and general manager at Smiths, explains that those design features, respectively, provide TSA officers with enhanced multiangle views of the contents and average-atomic-number data, which further differentiate benign materials from explosives.
If someone were driven to try to conceal an explosive weapon in a checked bag, it's quite likely that trace quantities of the explosive would adhere to the bag's contents and other surfaces. For that reason, TSA officers collect sample swabs from bags deemed suspicious on the basis of CT scanning and analyze those swabs with ion-mobility spectrometry (IMS), a highly sensitive detection method and another cornerstone of airport screening procedures.

In IMS measurements, a plume of sample ions, formed by exposing analyte molecules to a radioactive material or other ionization source, enters a drift tube and interacts with a gas, often at high pressure. Under the influence of an electric field, the ions are driven down the length of the tube, causing them to separate en route to a detector according to their mass, size, and shape. The method generates unique IMS signatures that can be used to detect many types of compounds. According to Smiths, its IMS instruments, which are used at many airports, can detect cyclotrimethylenetrinitramine (RDX), pentaerythritol tetranitrate (PETN), and several other explosives at the picogram level in a few seconds.
TSA officers use much the same screening technology for carry-on bags as they do for checked luggage—X-ray imaging and IMS-based trace analysis—but carry-ons present unique safety concerns. "Here, we are looking for components that can be fabricated into an explosive device after the checkpoint," Malotky explains. News agencies widely reported in August 2006 that Islamic terrorists arrested at that time in the U.K. were planning to smuggle liquid precursors to explosive organic peroxides through airport security. The terrorists were charged with the intention of using the substances to destroy trans-Atlantic aircraft in flight. A 2008 court ruling exonerated them of that particular charge, but the arrests and news of the thwarted plot led to immediate restrictions that are still enforced today on traveling with liquids in carry-on luggage.
In addition to metal detectors and the familiar benchtop X-ray and IMS tools used at passenger checkpoints, other types of detectors play key roles in ensuring air-travel safety. At some airports, passengers walk through trace-portal or "puffer" machines, which screen travelers for explosive residues. These instruments direct fine jets of air at passengers to dislodge nanogram specks of explosives that may be clinging to clothing, skin, or hair and analyze the samples automatically via IMS. These machines are not without their problems, however. Last week, several newspapers reported that TSA was stopping its use of the 207 puffer machines it had purchased because dust, humidity, and jet fumes made them unreliable.
TSA officers may also have two types of handheld instruments available for chemical detection. One of the tools is based on IMS. The other, made by ICx Technologies, based in Arlington, Va., capitalizes on large changes in fluorescence caused by binding of explosive analyte molecules to specially designed conjugated aromatic polymers. A key feature of those polymers, which were designed by Timothy M. Swager's research group at Massachusetts Institute of Technology, is their ability to quickly shuttle electronic excitations along the polymer backbone.
Aimée Rose, a senior scientist at ICx and a former Swager group member, explains that depending on the exact nature of the polymer and the analytes it binds, facile electronic communication either rapidly quenches the fluorescence process or enhances it. Fluorescence enhancement is the basis of ICx's handheld Fido PaxPoint, a device used by TSA to screen passengers and baggage for peroxides and other hazardous liquids with low part-per-billion sensitivity.
TO EFFICIENTLY detect weapons, explosives, and other dangerous items that may be hidden in clothing or concealed on a traveler's body, the security administration is exploring numerous whole-body imaging technologies. Instruments that record images made from low-intensity backscattered X-rays and millimeter-wave light are available commercially and are being used in several airport pilot studies. The machines can readily reveal hidden objects without physical contact. But because they render clothing invisible, they can also reveal passengers' contours in what could be embarrassing detail. Accordingly, TSA is exploring how to use computerized methods to ensure privacy and studying the public's acceptance of the technology. The agency is also studying imaging techniques with terahertz- and radio-frequency light, infrared light, and other types of radiation.
Meanwhile, TSA is collaborating with scientists in many labs to explore ways of enhancing detection of explosives. For example, at Pacific Northwest National Laboratory (PNNL), researchers are studying ways to improve procedures for collecting and concentrating trace levels of explosives to improve detection sensitivity and selectivity.
In one PNNL study, Shane Addleman and coworkers compared commercial IMS cloth sampling wipes with fiberglass wipes modified with organic self-assembled monolayers (SAMs) and evaluated the wipes' usefulness as sample collectors. Control experiments on common explosives such as trinitrotoluene (TNT) showed that collecting samples with the SAM-modified materials boosted IMS signals by a factor of three.

PNNL's Scott D. Harvey is also looking for improved ways of collecting trace-explosive samples. He has compared standard solid-phase microextraction fibers, which are used for concentrating analytes, with ones coated with a β-diketonate polymer. Gas chromatography-mass spectrometry analysis of spiked air samples showed that at a concentration of 3 ppt, TNT is easily detected by the coated fibers but below the detection limit of the control fibers. Likewise, the coated fibers gave a 10-fold enhancement in the signal for 2,4-dinitrotoluene, a TNT degradation product (J. Chromatogr., A 2008, 1213, 110).
"We're not messing around with the configuration of the instrument," says David A. Atkinson, PNNL's head of explosives detection. "We're just trying to make better sample collectors by finding materials with enhanced affinity for explosives."
IN OTHER STUDIES, PNNL scientists indeed have modified instrument configurations. By replacing the radioactive ionizer commonly found on IMS instruments with a corona-discharge source, the researchers boosted selectivity for explosives. Atkinson explains that in the presence of air, the discharge forms NOx- ions, which are highly electronegative. That means they hold on to their charge and do not interact strongly with other background molecules, and so the system remains blind to them. But not to nitro-explosives. Because of their polarity and electronegativity, these molecules readily form charged adducts with NO x- ions. As a result, those analytes stand out sharply from the background (Talanta 2008, 77, 249).
The high sensitivity, fast response time, and chemical specificity of mass spectrometers could make those tools useful explosives detectors that generate low rates of false positives. Yet their size, complexity, and sample preparation requirements have made them unlikely to be selected for general airport screening applications. At Purdue University, R. Graham Cooks's research group is working on instrument designs that might bring mass spectrometry to an airport near you.
Recently, the researchers designed and built an easy-to-use miniature mass spectrometer that is fitted with an ion-trap mass analyzer and operated with a desorption electrospray ionization (DESI) source. In the DESI method, ions are created by spraying an aqueous nebulized solvent at a surface. The shoe-box-sized mass spec sucks the ions into the instrument and generates a mass spectrum within a few seconds. The group reports that the miniature device can detect traces of numerous explosives present on paper, plastic, skin, and other surfaces at the subnanogram level without the need for sample preparation.

Moving from small to really small microfabricated devices also figures into the research programs of scientists studying explosives detection. Thomas G. Thundat of Oak Ridge National Laboratory and coworkers there and at Technical University of Denmark, in Lyngby, have designed a detector featuring micrometer-scale silicon bridges that can be heated quickly and controllably. The device functions as a fast-acting, self-cleaning probe that can distinguish adsorbed analyte molecules on the basis of their thermal profiles.
Advertisement
In proof-of-concept experiments, the researchers demonstrated that the microscale device can almost instantaneously differentiate vapors of explosive compounds from nonexplosive ones and can distinguish among common explosive analytes such as TNT, PETN, and RDX. The group notes that its micro-differential thermal analyzer can measure reproducible thermal response patterns within 50 milliseconds with a detection limit of 600 pg (Rev. Sci. Instrum. 2009, 80, 035102).
Scientists aren't short on ideas for detecting hazardous substances. "But just because a technology works well doesn't mean it will be implemented in airports," says John S. Verrico, a spokesman for the Department of Homeland Security. He explains that promising ideas may be superseded by other good ideas or they may be too expensive to implement on a wide scale.
"It's often hard to make the leap from prototype to product," Hallowell remarks. Sometimes things work perfectly in the laboratory, but then, in Malotky's words, "sobering operational reality" sets in. Unlike laboratories, airport environments aren't easily controlled. They are bustling places filled with noisy eateries and subject to temperature and humidity swings. A detector may exhibit outstanding sensitivity in a clean laboratory, but when presented with a sample in a frappuccino and French fry matrix, it may be tough to pick out the analyte at the part-per-billion level.
Nonetheless, the search for improved detection technology goes on. Even as scientists develop techniques to mitigate new types of threats, TSA is working to make security screening faster and less intrusive and to do away with the ban on liquids and the need to remove shoes. In future airports, passengers may be screened unknowingly by "stand-off" technologies that can spot suicide bombers' hidden weapons before they enter the terminal. Checkpoint-free airports may not open for business in the near future. But the technology to secure them is being developed today.
.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter