Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Homoallenylation Sans Separation
Organic Synthesis: Chiral reaction could be used to make natural products, drugs
by Stu Borman
October 19, 2009
| A version of this story appeared in
Volume 87, Issue 42
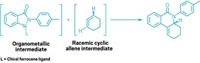
Researchers have devised the first enantioselective catalytic homoallenylation of aldehydes that produces β-allenols without requiring their tedious separation from 1,3-dienes, a common by-product of such reactions ( Angew. Chem. Int. Ed. , DOI: 10.1002/anie.200903647).
The work has implications for organic synthesis and drug discovery, as the homoallenyl alcohol product is a key building block in natural product and drug synthesis.
In 2002, synthetic organic chemist Patrick J. Guiry and coworkers at University College Dublin reported a series of chiral tridentate bis(oxazoline) ligands that helped direct product formation in chromium-catalyzed carbon-carbon bond-forming reactions. In the course of developing applications for the ligands, the researchers noticed that one of them catalyzed addition of a homoallene group (H2C=C=CH–) to benzaldehyde to produce 100% of the corresponding β-allenol without any by-product whatsoever, which is atypical. What’s more, the reaction boasted extremely high enantioselectivity—96% enantiomeric excess (ee).
By experimenting further with the ligand to assess its breadth of applicability, they were pleasantly surprised to find, in reactions on 10 aldehydes, that regioselectivities were 100% in nine cases (and 97% in the 10th), and ee’s were in the 90 to 98% range in eight cases (and 60% and 87% in two others).
“Previous work in the field has predominantly led to unsatisfactory mixtures of regioisomeric products,” Guiry says. Even in cases where they could be separated, enantioselectivities tended to be lower than in the new reactions, he notes.
The group doesn’t yet know the mechanism for the high regio- and enantioselectivities of the reactions. “That is the subject of current and future work,” Guiry says.
Yoshinori Yamamoto of Tohoku University, a carbonyl allylation and allenylation specialist, says the reaction “is a first-of-a-kind enantioselective transformation.” The enantioselectivities are high, but chemical yields are low, averaging around 50%, he notes. Guiry believes the yields can be improved.
In any case, homoallenylation “is very important for the synthesis of complicated molecules, and the new reaction will become a useful addition to the enantioselective repertoire,” Yamamoto concludes.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter