Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Materials
Fullerenes In A Box
Porphyrin cage sequesters massive aromatic guests
by Sarah Everts
March 21, 2011
| A version of this story appeared in
Volume 89, Issue 12
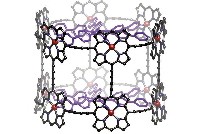
A self-assembling cube that can pack away massive aromatic guests such as coronene and fullerenes from the surrounding solution is being reported by chemist Jonathan R. Nitschke and colleagues at the University of Cambridge (Angew. Chem. Int. Ed., DOI: 10.1002/anie.201100193). The supramolecular cage has six walls, each composed of a modified porphyrin. The cubic cage’s eight corners are demarcated by eight iron atoms, which are held in place by ligands affixed to the porphyrin walls. Nitschke says this is the first cubic cage big enough to completely sequester large aromatic guests from the environment. Because the cage walls’ π-electron density sequesters C70 better than the more curved C60 incarnation, Nitschke says the cages could be useful for encapsulating or separating higher fullerenes from fullerene soot. He also sees applications in keeping reactive molecules or drugs sequestered until an appropriate signal permits their release. Next up, Nitschke says he hopes to do some metalloporphyrin-catalyzed chemistry in the caged environment that could be varied depending on which metals were placed into the porphyrin scaffold.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter