Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
Marking Sugar’s Spot
by Carmen Drahl
June 20, 2011
| A version of this story appeared in
Volume 89, Issue 25
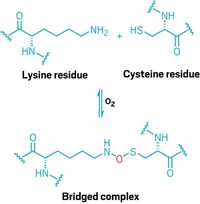
By using X-ray crystallography, researchers have learned how a sugar-transferring enzyme recognizes peptide sequences that trigger a common protein modification reaction—asparagine-linked glycosylation (Nature, DOI: 10.1038/nature10151). Saccharide attachment at asparagine’s side-chain nitrogen is important for protein folding, cell-cell communication, and more, but researchers know little about the attachment process. It requires either a serine or threonine to be two residues away from the asparagine to be modified, but it’s not clear how asparagine’s nitrogen, a poor nucleophile, gets activated for modification. To answer that question, Kaspar P. Locher, Christian Lizak, and coworkers at ETH Zurich crystallized a bacterial transferase together with a peptide substrate. They found that the hydroxyl-containing amino acid helps the enzyme recognize the substrate, but it doesn’t facilitate the reaction. That job belongs to an aspartate and a glutamate in the transferase, which juice up the asparagine by forming hydrogen bonds to its amide protons. The team says more structures need to be solved to fully understand the reaction.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter