Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Acid Catalyst spurs Chiral Addition
A Brønsted acid’s attachment to an alkene controls selectivity, but by a different pathway than metal catalysts
by Carmen Drahl
February 14, 2011
| A version of this story appeared in
Volume 89, Issue 7
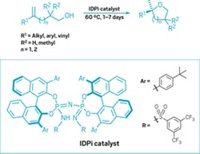
Add a new entry to the list of reactions that bulky proton donors can catalyze: asymmetric additions to carbon-carbon double bonds (Nature, DOI: 10.1038/nature09723). Researchers typically turn to chiral proton-donating Brønsted acids to functionalize carbonyl or other polar groups. Instead, F. Dean Toste and coworkers at the University of California, Berkeley, used a dithiophosphoric acid to turn dienes into chiral five-membered ring pyrrolidines and tetrahydrocarbazoles. The acid’s chiral counterion controls the reaction’s selectivity by covalently attaching to the diene, and it is ultimately ejected once the chiral five-membered ring forms. Metal catalysts can accomplish this type of reaction, but the UC Berkeley study provides proof of concept that acids help make chiral products from alkenes through a pathway that is distinct from the ones metal catalysts take, Toste explains. This pathway appears to avoid carbocation intermediates that typify textbook descriptions of acid reactions with double bonds, he adds. The UC Berkeley team is now working to apply the chemistry to a range of substrates.





Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter