Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Materials
Implantable Hydrogels Release Drugs, One At A Time
Drug Delivery: Gels could help doctors trigger multistep healing
by Katherine Bourzac
July 27, 2012
To avoid harmful side effects from protein drugs, some researchers have designed drug-loaded hydrogels to implant near diseased tissue. These gels could release large payloads of medication where and when it’s needed, without exposing healthy tissue. As an improvement to such gels, researchers have designed one that can release multiple drugs, one at a time, when treated with specific molecules (J. Am. Chem. Soc., DOI: 10.1021/ja305238a).
Releasing drugs at different times could allow doctors to treat a complex disease as each stage develops, or to aid a multi-step healing process. For example, says bioengineer Yong Wang of the University of Connecticut, Storrs, blood-vessel regeneration goes through several stages, each initiated by unique growth factors. Doctors could place a gel near damaged cardiac tissue during open-heart surgery, he says, so that after surgery, they could trigger the gel to release growth factors appropriate to stages of the regeneration process.
Some current drug-releasing gels can deliver medication in response to signals from outside the body, such as applied electric or magnetic fields. But these implants can’t dispense more than a single drug, Wong notes.
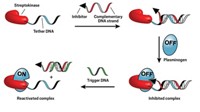
To produce gels that could release multiple drugs selectively, Wang and his team added DNA aptamers to gel mixtures. They picked aptamers because these short strands of nucleic acids bind protein drugs with high affinity, allowing the team to load the gel with their drugs of choice. Then, to release the proteins, all they would need is a short DNA strand that could bind to the protein-binding site of the aptamer. Once it did so, the aptamer would release its protein. By injecting different DNA sequences into the gel, the scientists could trigger delivery of specific drugs.
To test the idea, the researchers loaded a hydrogel with two heart-healing proteins: vascular endothelial growth factor (VEGF) and platelet-derived growth factor (PDGF).
To do so, the researchers coated polystyrene microparticles with aptamers for the two proteins, which other researchers had designed. They next incubated the particles in a solution of the two drugs for a couple of hours to allow the proteins to bind. To form the gel, the scientists then mixed these drug-coated particles with agarose, poured the mixture into cylindrical molds, and allowed the gel to set. (In future studies, Wang plans to replace polystyrene with a biodegradable material.) Each gel contained about a nanogram of each protein. Finally, the researchers designed complementary sequences for each aptamer binding site, each tens of nucleotides long, to trigger drug release.
To test how well the gels released drugs on cue, the researchers placed them in vials of buffer at body temperature and added one of the complementary sequences. They periodically collected and analyzed the fluid using enzyme-linked immunosorbent assay, better known as ELISA, to detect the freed proteins. After an hour, about 10% of the VEGF escaped when incubated with its respective triggering sequence, while about 8% of the PDGF released with the other sequence. As a result, the gels released tens of picograms of each protein in that hour. The team also found that they could release one drug from the gel after first dispensing the other.
Michael Cima, an engineer at Massachusetts Institute of Technology who also designs drug-releasing implants, sees promise in the aptamer approach. But he points out that for the gels to be effective in the body, they must release very large amounts of protein—clinically approved protein drugs tend to require large doses on the order of hundreds of milligrams.
Wang agrees and says his group is optimizing the gels to increase the dose by an order of magnitude. He thinks he can tweak the hydrogel structure to make it easier for the freed proteins to escape.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter